TYPE: Natural History Note![]()
RECEIVED 19 January 2026
ACCEPTED 27 January 2026
ONLINE EARLY 24 March 2026
PUBLISHED 30 March 2026
Abstract
In the spot-billed pelican (Pelecanus philippensis), sexes are phenotypically monomorphic, though a slight size difference is suspected. This study aimed to determine the extent of sexual size dimorphism (SSD) by examining 10 morphometric characters in 41 adult pelican carcasses (18 males, 23 females) collected at the Kokkare-Bellur Community Reserve, India, between 2018 and 2022. The pelicans were surgically sexed for confirmation. Males were found to be significantly larger than females in nine of the 10 measurements. Notably, culmen length showed a clear distinction with no overlap; lengths less than 320 mm were classified as female and more than 337 mm as male. Comparison of wild pairs through digital photographs confirmed this dimorphism. Furthermore, unique spot patterns on the beak of each individual were discovered, which can facilitate non-invasive individual identification for future behavioral investigations.
Keywords: Beak spots, culmen length, digital photographs, Kokkare-Bellur, morphometrics, non-invasive monitoring, phenotypic monomorphism
In the avian world, over 50% of species are considered monomorphic, with sexes being indistinguishable in plumage (Griffiths et al., 1998). However, sexual size dimorphism (SSD) often exists where reproductive roles are segregated, frequently associated with sex-specific behaviors and life-history strategies (Liker et al., 2021; Fairbairn et al., 2007). While males are often larger than females in birds, notable exceptions exist, such as Falconiformes, Strigiformes, and Charadriiformes (Fairbairn, 1997). The larger female may be advantageous for incubating larger clutches, defending nests, or competing with partners, like in Jacanas (Emlen & Wrege, 2004). In the family Pelecanidae, although sexes generally appear alike, male-biased SSD is known to occur. For instance, it has been determined in the American white pelican (Pelecanus erythrorhynchos) (Dorr et al., 2005). In the case of Spot-billed pelican P. philippensis, sexes are again reported to be alike (Ali & Ripley, 1978), though males may be larger.
The Spot-billed pelican is among the smallest of the Old World pelicans and is a tree-nesting species. Its historical range across South Asia has contracted significantly, and it is now confined to smaller pockets in India, Sri Lanka, Thailand, Cambodia, and Sumatra (BirdLife International, 2017). Due to a declining population, the species is classified as Near Threatened (BirdLife International, 2017). Southern India hosts 21 known breeding colonies, primarily in coastal regions (Subramanya, 2006), though additional unreported breeding sites may exist. An exception is the inland pelicanry at Kokkare-Bellur in Karnataka, believed to be one of the oldest nesting sites in southern India, where pelicans have nested within the village for over 300 years (Manu & Jolly, 2000; Neginhal, 1977).
In 2017-18, this site witnessed a large-scale mortality of adult pelicans, which was potentially linked to gastrointestinal parasitic infections (Kumar et al., 2019). This alarming event prompted the Karnataka State Forest Department to initiate a long-term monitoring project in 2018. Following this, over the course of four breeding seasons (2018-22), the pelicans that died were first measured for their morphometry, and then their sexes were identified through the surgical post-mortem. This note utilizes these specimens to provide the first detailed quantification of SSD in the spot-billed pelican using morphometric data. It further explores the reliability of using these findings, particularly culmen length, and digital photography to sex pelicans in the wild, thereby providing a valuable, non-invasive tool for conservation research on this Near Threatened species.
Morphological information was recorded from these dead pelicans for 10 characteristics, including head length, culmen length, culmen width between nares, culmen width maximum, culmen depth, tarsus length, tarsus width, mid-toe length, measured to the nearest 0.02 mm using Vernier caliper (Aerospace 300 mm). Tail and wing chord length were measured to the nearest 1 mm using a ruler (Figure 1).

Figure 1. Linear morphological measurements of 10 characteristics were taken of adult spot-billed pelicans at the Kokkare-Bellur Community Reserve, India. Tail length is not marked in the image. It was measured as part of the total length by gently placing the base of the ruler against the base of the middle pair of tail feathers.
All specimens were confirmed to be breeding adults through surgical examination of the reproductive organs during post-mortem. Body mass was intentionally excluded from the analysis as the specimens were in poor physiological condition due to parasitic infections, which would have significantly skewed this variable. However, the skeletal and keratinized structures measured are inherently stable and not subject to the rapid atrophy seen in soft tissues. To assess field applicability, culmen lengths were compared using calibrated ratios derived from digital photographs of known breeding pairs. Digital photographs of wild pelican pairs were obtained using a Canon PowerShot SX60HS (zoom range: 3.8-247mm) after a pair’s gender was determined following a mating event. The male mounts over the female and holds her neck for a brief period of time while they mate. This was an essential step since the spot-billed pelican builds its nests in clusters, which means that multiple nests will be joined to form a single platform, making it difficult to tell which sexes and pairings they belong to. To minimize measurement errors, we implemented the following controls: Only photographs where both individuals’ beaks were in the same plane and at similar angles relative to the camera were selected; Only photographs showing both pair members in lateral profile with beaks parallel to the camera plane were used. A total of 36 photographs (from an initial pool of >80 images) met these stringent selection criteria for image processing in Adobe Photoshop CS6 to draw lines and measure line length in pixels. Sex-wise differences were evaluated using non-parametric statistics (Mann-Whitney U test), and sexual size dimorphism was quantified using a mean difference index as proposed by Delestrade (2001) to determine the degree of dimorphism in each morphological character.
Mean difference index (MDI)=
(Mean character measurement for female)/(Mean character measurement for male) × 100
The comparison of morphological characters in 18 male and 23 female spot-billed pelicans showed significant differences in nine of the 10 characters measured. SSD ranged from 4-14% between sexes, with the mean difference index (MDI) exceeding 80% for all characters. Males were substantially larger than females, particularly in head and culmen length (14% larger) and tarsus width and length (10-13% larger) (Table 1).
Although the means for nine measurements were larger for males, there was overlap in the range of values for all characters except for culmen length (Table 1). The measurement of culmen length proved to be a definitive diagnostic tool; the culmen of males was consistently longer than 337 mm, while the culmen of females was always less than 320 mm (Figure 3).
The reliability of using culmen length to sex pelicans in the wild was tested by analyzing 36 digital photographs of known pairs (Figure 2). The ratio of male to female culmen length, calculated from these images, confirmed that males possess a longer beak, with an average ratio of 1.19 ± 0.09 (SD).
To enhance field identification methods, other physical attributes were compared. This investigation revealed no reliable differences between sexes in the presence of a crest, plumage coloration, or eye-patch color. Both sexes displayed a crest, over 90% of individuals had white plumage, and the majority of both males and females exhibited pale yellow eye-patches. However, the study revealed that the spot patterns on the beak were unique to each individual. These patterns, varying from triangular and alphabet-shaped spots to continuous lines formed by converging spots, serve as a natural marker (Figure 4). The variations in beak spot patterns could provide a method for identifying individual birds, subject to for further scientific investigation.
In the family Pelecanidae, which contains 8 species in the genus Pelecanus worldwide, information on SSD is scanty. Most of the available information on morphometry is based on a handful of museum specimens, which suggests that males are larger than females (Marchant & Higgins, 1990; Ali & Ripley, 1978). Our results clearly show that in spot-billed pelicans, males are larger. Male-biased SSD is a common attribute seen in waterfowl (Székely et al., 2007). The advantages of males being larger have been explained with respect to contests over mates or resources, mate preferences by the opposite sex, and resilience to temporary food shortages (Székely et al., 2007). However, the reasons for the male-biased SSD in spot-billed pelicans are still unclear, as there is no information available on the sex differences in breeding behavior and ecology.
Significant SSD was found in nine of the 10 morphological characters considered in this study (Table 1). However, culmen length alone showed clear distinction with no overlap, which can be used to determine the sex of adult spot-billed pelicans. A similar lack of overlap in culmen length was utilized to successfully determine the sex of the American white pelican (P. erythrorhynchos) (Dorr et al., 2005). While our results provide a clear threshold for the Kokkare-Bellur population, it remains to be seen if this striking difference holds across other geographically separated populations or if these specific values are population-specific. Currently, no other studies have been conducted in India or other regions within the species’ range to provide a comparative baseline. Simple linear measurement of the beak has been used to predict sex in many other waterbirds (e.g., Riordan & Johnston, 2013; Devlin et al., 2004; Murata et al., 1988). The size of the culmen may be affected by both inter-

Figure 2. A nesting pair of spot-billed pelican at Kokkare-Bellur Community Reserve, southern India. The male is larger with a long beak than the female during a courting incident. L1 (female) and L2 (male) depict culmen length considered during measurement in AdobePhotoshop CS6.
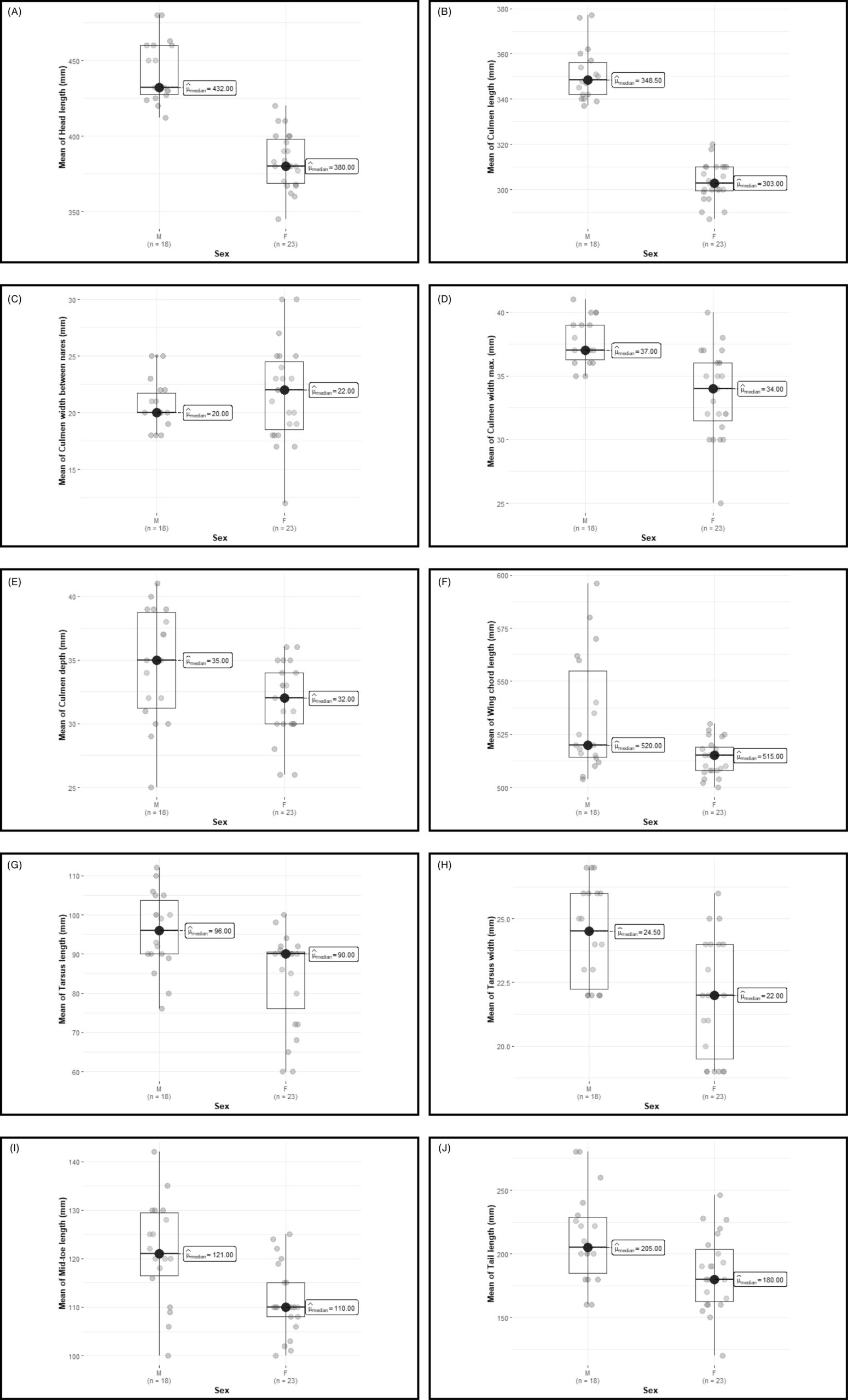
Figure 3. Box plots illustrating the variation in morphological characters between male and female spot-billed pelican. Panels represent: (A) head length, (B) culmen length, (C) culmen width between nares, (D) maximum culmen width, (E) culmen depth, (F) wing chord length, (G) tarsus length, (H) tarsus width, (I) middle toe length, and (J) tail length. The thick horizontal line inside each box indicates the median, the box represents the interquartile range (IQR), and the whiskers denote the standard deviation. Grey circles represent individual data points. Significant differences between sexes were observed for all morphological characters (P < 0.05), except for culmen width between nares, which did not show a significant difference (P = 0.474).
and intrasexual selection (De Marchi et al., 2012). The longer beak may allow males to produce loud clapping sounds to attract females or to threaten potential predators. While bill-clapping is a recognized display in the genus Pelecanus, the specific relationship between culmen dimensions and acoustic properties in spot-billed pelicans warrants further bioacoustic investigation. During mating, when the male mounts the female, males with longer beaks may be at an advantage when they use their beaks to grasp the female’s neck (Gokula, 2011). Longer beaks in males may also be a result of greater fitness through sexual selection, as they allow males to more efficiently harvest food resources (Bildstein, 1987) or gather nesting material (Urfi & Kalam, 2006). The culmen is also likely to be more useful for identifying the sex of pelican carcasses, as it degrades more slowly than softer tissues (Dorr et al., 2005).
Table 1. Comparison between male and female adult breeding spot-billed pelicans, based on 10 characteristics. Mean difference index (MDI) calculated as (mean characteristic measurement for female/mean characteristic measurement for male) × 100.
| Characteristics | Male n = 18 Mean ± SD (range) | Female n = 23 Mean ± SD (range) | Mann-Whitney U statistic | MDI (%) |
|---|---|---|---|---|
| Head length (mm) | 442.6 ± 20.6 (412–480) | 383.4 ± 18.2 (345–420) | 1.50 | 86.6 |
| Culmen length (mm) | 350.8 ± 11.7 (337–377) | 303.3 ± 8.4 (287–320) | 0.00 | 86.4 |
| Culmen width between nares (mm) | 20.6 ± 2.0 (18–25) | 21.6 ± 4.3 (12–30) | 234.00 | 95.3 |
| Culmen width max. (mm) | 37.7 ± 1.8 (35–41) | 33.6 ± 3.4 (25–40) | 62.00 | 89.1 |
| Culmen depth (mm) | 34.6 ± 4.5 (25–41) | 31.7 ± 2.8 (26–36) | 125.00 | 91.6 |
| Tarsus length (mm) | 95.6 ± 10.0 (76–112) | 84.1 ± 11.8 (60–100) | 102.50 | 87.9 |
| Tarsus width (mm) | 24.3 ± 1.9 (22–27) | 21.8 ± 2.2 (19–26) | 86.00 | 89.7 |
| Mid-toe length (mm) | 121.4 ± 10.6 (100–142) | 111.2 ± 7.0 (100–125) | 92.50 | 91.5 |
| Tail length (mm) | 212.7 ± 36.0 (160–280) | 185.5 ± 29.6 (120–246) | 118.00 | 87.2 |
| Wing chord length (mm) | 533.4 ± 28.0 (504–596) | 513.8 ± 8.5 (500–530) | 117.00 | 96.3 |

Figure 4. Variation in beak spot patterns of spot-billed pelican pairs photographed during breeding time at Kokkare-Bellur Community Reserve. A-I are 9 different pairs, and the black circles in the beak depict their unique spots.
Sexing birds using digital photographs has been tested before and proved to be a highly reliable, non-invasive method to sex individuals (Williams et al., 2020; Weckauf & Handschuh, 2011; Urfi & Kalam, 2006; Cheong et al., 2007). Such non-invasive techniques are particularly valuable for morphometric studies on wild, free-ranging animals on the Indian subcontinent, where capturing may not be feasible (Mahendiran et al., 2018). This method has also helped to understand social interactions of species, such as larger painted stork (Mycteria leucocephala) males, which acquired better nesting sites over territorial winning from smaller conspecifics (Urfi & Kalam, 2006). Males of white ibis (Eudocimus albus) during nesting were found to be more agile in stealing prey than females as they were more efficient in intimidating others (Frederick, 1985). Foraging efficiency when compared between male and female Oriental white storks (Ciconia boyciana) revealed sex-based differences in foraging habitats and methods of foraging (Sung et al., 2009).
Likewise, this study can be useful in a easier identification of sex in the field for studying sex-specific social, nesting, and foraging behaviors, understanding energetics, parental investments, and effects of differential sex ratio in the population. Future research should specifically leverage these sexing techniques to explore the ecological impacts on mortality across varying habitats. In particular, at sites like Kokkare Bellur, comparing the survival rates of spot-billed pelicans in man-made wetlands versus their natural habitats could reveal if sex-specific vulnerabilities exist. Understanding these mortality drivers is critical for the long-term conservation of the species.
Once a mating pair has been identified in the field, the male and female can be distinguished by the differences in their beak length. As the pairs are rarely observed together during the breeding season apart from the mating and nest-building events, researchers should develop methods to mark the individuals based on their unique beak spots. This method will aid in conducting detailed scientific investigations on their nesting behavior.
Acknowledgement
This study forms part of a long-term monitoring programme on the Spot-billed Pelican funded by the Karnataka State Forest Department. I thank my doctoral supervisor, Dr. R. Suresh Kumar, for initiating the study, for his integral involvement throughout the project, and for providing guidance during the preparation of this manuscript. I also acknowledge Dr. Sathish (Senior Veterinary Officer, Animal Husbandry Department, Maddur) and veterinary officers from Bengaluru and Mysuru for providing information on the gender of the adult pelicans. I thank Mr. Manjunathan J. for sharing pelican photographs and for assistance with figure preparation and data analysis. I am grateful to Dr. Greg Johnston, Ms. Amarjeet Kaur, and Mr. Mohit Mudliar for their constructive comments on an earlier version of the manuscript.
CONFLICT OF INTEREST
The author declares no conflict of interest.
DATA AVAILABILITY
Data is available on online free public source at Figshare. (https://doi.org/10.6084/m9.figshare.31083748)
AUTHORS’ CONTRIBUTION
AM performed all roles, including conceptualization, investigation, analysis, and writing.
ETHICS STATEMENT
All procedures involving animal handling were conducted in compliance with the Wildlife (Protection) Act, 1972 of India. Permission for handling of Spot-billed Pelicans was granted by the Principal Chief Conservator of Forests (PCCF), Karnataka State Forest Department. All veterinary examinations and treatments were carried out by authorized veterinarians deputed by the Karnataka Forest Department. The study did not involve invasive sampling. The research adhered to national wildlife welfare regulations and ethical standards for the handling of protected species, and complied with the principles of the Convention on Biological Diversity and CITES.
DECLARATION OF THE USE OF GENERATIVE AI
The authors used Gemini 2 Pro for language improvement and paraphrasing during the preparation of this manuscript. The authors thoroughly reviewed and edited all content generated using this tool and take full responsibility for the accuracy, integrity, and originality of the content of the publications.
Edited By
Mewa Singh
University of Mysore, Mysore, India.
*CORRESPONDENCE
Aksheeta Mahapatra
✉ mail.aksheeta95@gmail.com
CITATION
Mahapatra, A. (2026). Spotting the difference: Sexual size dimorphism and individual identification in the Spot-billed Pelican Pelecanus philippensis. Journal of Wildlife Science, 3(1), 30-34. https://doi.org/10.63033/JWLS.NABN4710
FUNDING
This study was supported by the Karnataka State Forest Department, Mysuru Wildlife Division, under project approval number PCCF (WL)/E2/ CR-28/2019–20.
COPYRIGHT
© 2026 Mahapatra. This is an open-access article, immediately and freely available to read, download, and share. The information contained in this article is distributed under the terms of the Creative Commons Attribution License (CC BY 4.0), allowing for unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited in accordance with accepted academic practice. Copyright is retained by the author(s).
PUBLISHED BY
Wildlife Institute of India, Dehradun, 248 001 INDIA
PUBLISHER'S NOTE
The Publisher, Journal of Wildlife Science or Editors cannot be held responsible for any errors or consequences arising from the use of the information contained in this article. All claims expressed in this article are solely those of the author(s) and do not necessarily represent those of their affiliated organisations or those of the publisher, the editors and the reviewers. Any product that may be evaluated or used in this article or claim made by its manufacturer is not guaranteed or endorsed by the publisher.
Ali, S. & Ripley, S. D. (eds.), (1978). Handbook of the birds of India and Pakistan, together with those of Bangladesh, Nepal, Bhutan and Sri Lanka. Oxford University Press, London, New York, 1(2).
Bildstein, K. L. (1987). Energetic Consequences of Sexual Size Dimorphism in White Ibises (Eudocimus albus). The Auk, 104, 771–775. https://doi.org/10.1093/auk/104.4.771
BirdLife International. (2017). Threatened Birds of Asia: The BirdLife International Red Data Book. BirdLife International, Cambridge.
Cheong, S., Sung, H-C. & Park, S-R. (2007). A new method for sexing Oriental White Storks. Journal of Field Ornithology, 78(3), 329–333. https://doi.org/10.1111/j.1557-9263.2007.00112.x
De Marchi, G., Fasola, M., Chiozzi, G., Bellati, A. & Galeotti, P. (2012). Sex Discrimination of Crab Plovers (Dromas ardeola) by Morphometric Traits. Waterbirds, 35(2), 332–337. https://doi.org/10.1675/063.035.0214.
Delestrade, A. (2001). Sexual Size Dimorphism and Positive Assortative Mating in Alpine Choughs (Pyrrhocorax graculus). The Auk, 118(2), 553–556. https://doi.org/10.1093/auk/118.2.553
Devlin, C. M., Diamond, A. W. & Saunders, G. W. (2004). Sexing Arctic Terns in the field and laboratory. Waterbirds, 27(3), 314–320. https://doi.org/10.1675/1524-4695(2004)027[0314:SATITF]2.0.CO;2
Dorr, B., King, D. T., Harrel, J. B., Gerard, P. & Spalding, G. (2005). The Use of Culmen Length to Determine Sex of the American White Pelican. Waterbirds, 28(1), 102–106. https://doi.org/10.1675/1524-4695(2005)28[102:TUOCLT]2.0.CO;2
Emlen, S. T. & Wrege, P. H. (2004). Size Dimorphism, Intrasexual Competition, and Sexual Selection in Wattled Jacana (Jacana Jacana), A Sex-Role-Reversed Shorebird in Panama. The Auk, 121(2), 391-403. https://doi.org/10.1093/auk/121.2.391
Fairbairn, D. J., Blanckenhorn, W. U. & Székely, T. (eds.), (2007). Sex, Size and Gender Roles: Evolutionary Studies of Sexual Size Dimorphism. Oxford University Press, Oxford, New York. https://doi.org/10.1093/acprof:oso/9780199208784.001.0001
Fairbairn, D. J. (1997). Allometry for Sexual Size Dimorphism: Pattern and Process in the Coevolution of Body Size in Males and Females. Annual Review of Ecology and Systematics, 28, 659–687. https://doi.org/10.1146/annurev.ecolsys.28.1.659
Frederick, P. (1985). Intraspecific food piracy in White Ibises. Journal of Field Ornithology, 56, 413–414.
Gokula, V. (2011). Breeding biology of the Spot-billed Pelican (Pelecanus philippensis) in Karaivetti Bird Sanctuary, Tamil Nadu, India. Chinese Birds, 2(2), 101–108. https://doi.org/10.5122/cbirds.2011.0013
Griffiths, R., Double, M. C., Orr, K. & Dawson, R. J. G. (1998). A DNA test to sex most birds. Molecular Ecology, 7, 1071–1075. https://doi.org/10.1046/j.1365-294x.1998.00389.x
Kumar, S., Periyasamy, A., Rao, N. V. R., Sunil, S. S., Kumara, H. N., Sundaraj, P., Chidananda, G. & Sathish. A. (2019). Multiple infestations of gastrointestinal parasites – Probable cause for high mortality of Spot-billed Pelican (Pelecanus philippensis) at Kokrebellur Community Reserve, India. The International Journal for Parasitology: Parasites and Wildlife, 9, 68–73. https://doi.org/10.1016/j.ijppaw.2019.04.001
Liker, A., Bókony, V., Pipoly, I., Lemaître, J-F., Gaillard, J-M., Székely, T. & Freckleton, R. P. (2021). Evolution of large males is associated with female-skewed adult sex ratios in amniotes. Evolution, 75(7), 1636–1649. https://doi.org/10.1111/evo.14273
Mahendiran, M., Parthiban, M., Azeez, P. A., & Nagarajan, R. (2018). In situ measurements of animal morphological features: A non-invasive method. Methods in Ecology and Evolution, 9(3), 613-623. https://doi.org/10.1111/2041-210X.12898
Manu, K. & Jolly, S. (2000). Pelicans and People: The Two-Tier Village of Kokkare Bellur, Karnataka, India. Kalpavriksh and International Institute of Environment and Development.
Marchant, S. & Higgins, P. J. (eds.), (1990). Handbook of Australian, New Zealand and Antarctic Birds. Ratites to Ducks. Oxford University Press, Melbourne. 1(1).
Murata, K., Miyashita, M., Nagase, K., Komya, T. & Matushima, K. (1988). Sex determination in the Eastern white stork, Ciconia c. boyciana, by bill measurements and discriminant analysis. Journal of Japanese Association of Zoos and Aquariums, 30, 43–47.
Neginhal, S. G. (1977). Discovery of Pelicanry in Karnataka. Journal of Bombay Natural History Society, 74, 169–170.
Riordan, J. & Johnston. G. (2013). Morphological Sex Determination in Black-Faced Cormorants (Phalacrocorax fuscescens). Waterbirds, 36(1), 94–101. https://doi.org/10.1675/063.036.0114
Subramanya, S. (2006). Pelicans bounce back. World Birdwatch, 28(4).
Sung, H-C., Kim, J. H., Cheong, S-W., Kim, S-K., Jo, J-Y., Cheong, M-R., Choi, Y-S. & Park, S-R. (2009). A case study on foraging behavior of Oriental White Storks (Ciconia boyciana) in the variation of prey density and water depth. Korean Society of Environmental Biology, 27, 155–163.
Székely, T., Lislevand, T. & Figuerola, J. (2007). Sexual size dimorphism in birds. In: Fairbairn, D. J., Blanckenhorn, W. U. & Székely, T. (eds.). Sex, size and gender roles: evolutionary studies of sexual size dimorphism. Oxford University Press, Oxford, New York. pp.27-37.
Urfi, A. J. & Kalam. A. (2006). Sexual Size Dimorphism and Mating Pattern in the Painted Stork (Mycteria leucocephala). Waterbirds, 29(4), 489–496. https://doi.org/10.1675/1524-4695(2006)29[489:SSDAMP]2.0.CO;2
Weckauf, R. & Handschuh. M. (2011). A method for identifying the sex of lesser adjutant storks Leptoptilos javanicus using digital photographs. Cambodian Journal of Natural History, 1, 23–28.
Williams, H. M., Wilcox, S. B. & Patterson, A. J. (2020). Photography as a tool for avian morphometric measurements. Journal of Ornithology, 161, 333–339. https://doi.org/10.1007/s10336-019-01728-w
Edited By
Mewa Singh
University of Mysore, Mysore, India.
*CORRESPONDENCE
Aksheeta Mahapatra
✉ mail.aksheeta95@gmail.com
CITATION
Mahapatra, A. (2026). Spotting the difference: Sexual size dimorphism and individual identification in the Spot-billed Pelican Pelecanus philippensis. Journal of Wildlife Science, 3(1), 30-34. https://doi.org/10.63033/JWLS.NABN4710
FUNDING
This study was supported by the Karnataka State Forest Department, Mysuru Wildlife Division, under project approval number PCCF (WL)/E2/ CR-28/2019–20.
COPYRIGHT
© 2026 Mahapatra. This is an open-access article, immediately and freely available to read, download, and share. The information contained in this article is distributed under the terms of the Creative Commons Attribution License (CC BY 4.0), allowing for unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited in accordance with accepted academic practice. Copyright is retained by the author(s).
PUBLISHED BY
Wildlife Institute of India, Dehradun, 248 001 INDIA
PUBLISHER'S NOTE
The Publisher, Journal of Wildlife Science or Editors cannot be held responsible for any errors or consequences arising from the use of the information contained in this article. All claims expressed in this article are solely those of the author(s) and do not necessarily represent those of their affiliated organisations or those of the publisher, the editors and the reviewers. Any product that may be evaluated or used in this article or claim made by its manufacturer is not guaranteed or endorsed by the publisher.
Ali, S. & Ripley, S. D. (eds.), (1978). Handbook of the birds of India and Pakistan, together with those of Bangladesh, Nepal, Bhutan and Sri Lanka. Oxford University Press, London, New York, 1(2).
Bildstein, K. L. (1987). Energetic Consequences of Sexual Size Dimorphism in White Ibises (Eudocimus albus). The Auk, 104, 771–775. https://doi.org/10.1093/auk/104.4.771
BirdLife International. (2017). Threatened Birds of Asia: The BirdLife International Red Data Book. BirdLife International, Cambridge.
Cheong, S., Sung, H-C. & Park, S-R. (2007). A new method for sexing Oriental White Storks. Journal of Field Ornithology, 78(3), 329–333. https://doi.org/10.1111/j.1557-9263.2007.00112.x
De Marchi, G., Fasola, M., Chiozzi, G., Bellati, A. & Galeotti, P. (2012). Sex Discrimination of Crab Plovers (Dromas ardeola) by Morphometric Traits. Waterbirds, 35(2), 332–337. https://doi.org/10.1675/063.035.0214.
Delestrade, A. (2001). Sexual Size Dimorphism and Positive Assortative Mating in Alpine Choughs (Pyrrhocorax graculus). The Auk, 118(2), 553–556. https://doi.org/10.1093/auk/118.2.553
Devlin, C. M., Diamond, A. W. & Saunders, G. W. (2004). Sexing Arctic Terns in the field and laboratory. Waterbirds, 27(3), 314–320. https://doi.org/10.1675/1524-4695(2004)027[0314:SATITF]2.0.CO;2
Dorr, B., King, D. T., Harrel, J. B., Gerard, P. & Spalding, G. (2005). The Use of Culmen Length to Determine Sex of the American White Pelican. Waterbirds, 28(1), 102–106. https://doi.org/10.1675/1524-4695(2005)28[102:TUOCLT]2.0.CO;2
Emlen, S. T. & Wrege, P. H. (2004). Size Dimorphism, Intrasexual Competition, and Sexual Selection in Wattled Jacana (Jacana Jacana), A Sex-Role-Reversed Shorebird in Panama. The Auk, 121(2), 391-403. https://doi.org/10.1093/auk/121.2.391
Fairbairn, D. J., Blanckenhorn, W. U. & Székely, T. (eds.), (2007). Sex, Size and Gender Roles: Evolutionary Studies of Sexual Size Dimorphism. Oxford University Press, Oxford, New York. https://doi.org/10.1093/acprof:oso/9780199208784.001.0001
Fairbairn, D. J. (1997). Allometry for Sexual Size Dimorphism: Pattern and Process in the Coevolution of Body Size in Males and Females. Annual Review of Ecology and Systematics, 28, 659–687. https://doi.org/10.1146/annurev.ecolsys.28.1.659
Frederick, P. (1985). Intraspecific food piracy in White Ibises. Journal of Field Ornithology, 56, 413–414.
Gokula, V. (2011). Breeding biology of the Spot-billed Pelican (Pelecanus philippensis) in Karaivetti Bird Sanctuary, Tamil Nadu, India. Chinese Birds, 2(2), 101–108. https://doi.org/10.5122/cbirds.2011.0013
Griffiths, R., Double, M. C., Orr, K. & Dawson, R. J. G. (1998). A DNA test to sex most birds. Molecular Ecology, 7, 1071–1075. https://doi.org/10.1046/j.1365-294x.1998.00389.x
Kumar, S., Periyasamy, A., Rao, N. V. R., Sunil, S. S., Kumara, H. N., Sundaraj, P., Chidananda, G. & Sathish. A. (2019). Multiple infestations of gastrointestinal parasites – Probable cause for high mortality of Spot-billed Pelican (Pelecanus philippensis) at Kokrebellur Community Reserve, India. The International Journal for Parasitology: Parasites and Wildlife, 9, 68–73. https://doi.org/10.1016/j.ijppaw.2019.04.001
Liker, A., Bókony, V., Pipoly, I., Lemaître, J-F., Gaillard, J-M., Székely, T. & Freckleton, R. P. (2021). Evolution of large males is associated with female-skewed adult sex ratios in amniotes. Evolution, 75(7), 1636–1649. https://doi.org/10.1111/evo.14273
Mahendiran, M., Parthiban, M., Azeez, P. A., & Nagarajan, R. (2018). In situ measurements of animal morphological features: A non-invasive method. Methods in Ecology and Evolution, 9(3), 613-623. https://doi.org/10.1111/2041-210X.12898
Manu, K. & Jolly, S. (2000). Pelicans and People: The Two-Tier Village of Kokkare Bellur, Karnataka, India. Kalpavriksh and International Institute of Environment and Development.
Marchant, S. & Higgins, P. J. (eds.), (1990). Handbook of Australian, New Zealand and Antarctic Birds. Ratites to Ducks. Oxford University Press, Melbourne. 1(1).
Murata, K., Miyashita, M., Nagase, K., Komya, T. & Matushima, K. (1988). Sex determination in the Eastern white stork, Ciconia c. boyciana, by bill measurements and discriminant analysis. Journal of Japanese Association of Zoos and Aquariums, 30, 43–47.
Neginhal, S. G. (1977). Discovery of Pelicanry in Karnataka. Journal of Bombay Natural History Society, 74, 169–170.
Riordan, J. & Johnston. G. (2013). Morphological Sex Determination in Black-Faced Cormorants (Phalacrocorax fuscescens). Waterbirds, 36(1), 94–101. https://doi.org/10.1675/063.036.0114
Subramanya, S. (2006). Pelicans bounce back. World Birdwatch, 28(4).
Sung, H-C., Kim, J. H., Cheong, S-W., Kim, S-K., Jo, J-Y., Cheong, M-R., Choi, Y-S. & Park, S-R. (2009). A case study on foraging behavior of Oriental White Storks (Ciconia boyciana) in the variation of prey density and water depth. Korean Society of Environmental Biology, 27, 155–163.
Székely, T., Lislevand, T. & Figuerola, J. (2007). Sexual size dimorphism in birds. In: Fairbairn, D. J., Blanckenhorn, W. U. & Székely, T. (eds.). Sex, size and gender roles: evolutionary studies of sexual size dimorphism. Oxford University Press, Oxford, New York. pp.27-37.
Urfi, A. J. & Kalam. A. (2006). Sexual Size Dimorphism and Mating Pattern in the Painted Stork (Mycteria leucocephala). Waterbirds, 29(4), 489–496. https://doi.org/10.1675/1524-4695(2006)29[489:SSDAMP]2.0.CO;2
Weckauf, R. & Handschuh. M. (2011). A method for identifying the sex of lesser adjutant storks Leptoptilos javanicus using digital photographs. Cambodian Journal of Natural History, 1, 23–28.
Williams, H. M., Wilcox, S. B. & Patterson, A. J. (2020). Photography as a tool for avian morphometric measurements. Journal of Ornithology, 161, 333–339. https://doi.org/10.1007/s10336-019-01728-w




