TYPE: Short Communication![]()
RECEIVED 06 January 2025
ACCEPTED 17 March 2026
ONLINE EARLY 27 March 2026
PUBLISHED 30 March 2026
Abstract
We tracked two rescued spot-billed pelicans, Pelecanus philippensis, a hand-raised juvenile and a rehabilitated adult, using GPS telemetry for approximately two years after release from Kokkare-Bellur Community Reserve, southern India. Dynamic Brownian Bridge Movement Models revealed distinct age-dependent utilization distribution strategies. The juvenile exhibited progressive spatial expansion, from localized movements in the first year to distant wetlands in the second year. The adult dispersed immediately and remained largely within the Bengaluru–Mysuru corridor, with daily distance traveled increasing during the breeding season. Results demonstrate successful post-release dispersal and landscape use, highlighting the importance of wetland network connectivity for the conservation of the pelican.
Keywords: Dynamic Brownian Bridge Movement Model (dBBMM), Pelecanus philippensis, rehabilitation, space use, southern India, wetland connectivity.
Introduction
Wildlife rescue and rehabilitation are widely applied conservation interventions for injured, orphaned, or displaced birds, yet their effectiveness is rarely evaluated beyond short-term survival (Pyke et al., 2018; Hernandez, 2019). In this context, post-release monitoring is a powerful approach for assessing the functional outcomes of rehabilitation, including dispersal behavior, space use, and landscape integration (Bernardo et al., 2011; Raine et al., 2020; Rozsypalová et al., 2025). GPS telemetry studies on rehabilitated pelicans following oil spills have demonstrated altered movement strategies, increased dispersion, and reduced breeding investment relative to non-rehabilitated individuals, highlighting the need for long-term behavioural assessment of released birds (Lamb et al., 2018; Fiorello et al., 2021).
Pelicans are large, colonial, piscivorous waterbirds that function as top predators in aquatic ecosystems and are widely regarded as indicators of wetland health due to their sensitivity to changes in water quality, fish availability, and disturbance to breeding sites (del Hoyo et al., 1992; Amat & Green, 2009). Among the eight extant pelican species, the spot-billed pelican (Pelecanus philippensis), a tree-nesting species distributed across South and Southeast Asia, remains poorly studied (Kannan & Pandiyan, 2013; BirdLife International, 2017). In India, the species breeds at approximately 20 sites, primarily in southern states, and is classified as Near Threatened (BirdLife International, 2017) due to the loss of wetlands, human disturbance, and emerging disease threats (Kannan & Pandiyan, 2013). Despite its conservation importance, empirical information on its current population status and movements is limited, and more specifically, survival following rescue or rehabilitation efforts remains unknown.
Kokkare-Bellur Community Reserve represents one of the oldest and most important breeding sites for the spot-billed pelican in southern India. Alarmingly, between 2017 and 2018, this site experienced a mass mortality event involving more than 50 adult pelicans, later attributed to heavy gastrointestinal nematode infestations associated with degraded wetland conditions (Kumar et al., 2019). Similar mortality events linked to Contracaecum spp. infestations have since been reported from other pelican breeding and foraging sites in southern India (Durairajan, 2023; Mathews et al., 2025), underscoring growing concerns regarding adult survival and exposure to environmental stressors. These events prompted tracking fine-scale movements of rescued pelicans to understand their ranging behavior, wetland use, and evaluate the overall success of rehabilitation efforts.
Here, we present a case report of documenting spatio-temporal movements of two rescued spot-billed pelicans, a hand-raised juvenile and a rehabilitated adult, tracked for approximately two years using GPS telemetry following their release from Kokkare-Bellur site. This study offers a rare, detailed look into the individual-level movement ecology of this species, providing valuable data for future conservation and reintroduction programs.
Methods
This study was conducted as part of a five-year monitoring program of spot-billed pelican breeding ecology at Kokkare-Bellur Community Reserve, Karnataka. The site exemplifies a unique conservation model, wherein colonial waterbirds, primarily spot-billed pelicans and painted storks Mycteria leucocephala, nest within a human-inhabited village landscape. Nesting trees are distributed in scattered patches throughout residential areas, agricultural lands, and temple grounds rather than in an isolated tree patch in a wetland.
During monitoring, natural falls of pelican chicks and adults from nests were documented, attributed to sibling competition, adverse weather, fledging attempts, and illness. During the 2022–23 breeding season, a total of 13 chicks and two adults were rescued and rehabilitated, of which two individuals were selected for GPS tagging based on health status and growth parameters. A chick rescued at four days old on 16 January 2022 was hand-raised and released on 9 November 2022 (named Shimsha; body mass at tagging = 4865 g), while an adult pelican rescued in a dehydrated condition on 21 December 2022 received veterinary treatment and was released on 4 February 2023 (named Aadi; body mass at tagging = 4480g). Both pelicans were released near the village in proximity to the Shimsha River and were fitted with 33 g solar-powered GPS-GSM patagial transmitters (OrniTrack P33, Ornitela, Lithuania). Transmitters were programmed to record GPS locations at 10-minute intervals and transmit the data daily once. Movement data were collected from September 10, 2022, to August 31, 2024, for Shimsha (722 days) and from February 4, 2023, to February 28, 2025, for Aadi (755 days). For Shimsha, GPS data collection ended prematurely on August 31, 2024, after the transmitter detached and fell into the river; however, subsequent field observations confirmed that the bird retained the patagial tag and was repeatedly recorded flying, foraging, and hunting fishes, indicating normal movement and an apparently healthy condition.
For Shimsha, the data were partitioned into Year 1 (November 9, 2022, to November 9, 2023) and Year 2 (November 10, 2023, to August 31, 2024). The data were treated separately for each year to quantify annual changes in space use. For Aadi, data were partitioned into breeding and non-breeding seasons across two cycles. Breeding seasons were defined as October 2023 to April 2024 (Year 1) and October 2024 to February 2025 (Year 2) based on observed nesting activity and colony monitoring. Non-breeding seasons comprised the remaining months (February 2023 to September 2023 and May 2024 to September 2024).
Data were processed in R version 4.3.1 (R Core Team, 2024) using packages move (Kranstauber et al., 2024), raster (Hijmans et al., 2025), sp, (Pebesma et al., 2025a), and sf (Pebesma et al., 2025b). Prior to analysis, all GPS locations were imported into QGIS 3.28 and visually inspected for spatial outliers and implausible fixes. One erroneous outlier location was identified and removed from the dataset. There were no missing GPS fixes in the dataset. The daily distance traveled was calculated by summing the distances between consecutive locations within each 24-hour period. The daily distance travel data were summarized using median and range. For a visual representation, the daily distances traveled were plotted using a LOESS regression.
To quantify space use and identify core areas, we employed the Dynamic Brownian Bridge Movement Model (dBBMM), which estimates an animal’s probability of occurrence by incorporating the time and distance between consecutive locations and the animal’s movement variance (Kranstauber et al., 2012). The dBBMM was computed using the ‘brownian.bridge.dyn’ function in the package ‘move’ with a window size of 31 and a margin of 11 fixes to capture temporal variations in movement behavior. A location error of 20 m was specified, and a high-resolution grid (dimSize = 600) was used to ensure detailed contouring. From the resulting Utilization Distribution (UD) raster, we extracted isopleths for the 50% (core use area), 75% (primary use area), and 99% (overall utilization distribution) contours to quantify annual and seasonal differences in space use for each individual. Seasonal dBBMMs enabled the quantification of reproductive versus non-reproductive space-use patterns. Spatial outputs were visualized in QGIS 3.28, utilizing satellite imagery, to identify core wetlands and map movement patterns.
Results
Juvenile pelican (Shimsha) movement patterns
Shimsha was tracked for 722 days post-release, generating 104,112 GPS locations. Movement patterns were predominantly concentrated around the release location along the Shimsha River, with opportunistic exploratory movements to neighboring inland wetlands. A single long-distance movement to Mysuru (approximately 80 km from the release site) was also recorded, during which Shimsha visited multiple wetlands before returning to the core area.
During the first year (November 2022 to November 2023), Shimsha’s movements remained highly localized. The 50% core utilization area was very small (less than 1 km²). The 75% UD encompassed 1 km², whilst the 99% UD covered 32 km², capturing the full extent of exploratory movements along sections of the Shimsha River within 10 km of the release location and two neighbouring lakes.
In the second year (November 2023 to November 2024), Shimsha’s core areas shifted spatially and expanded substantially. The 50% UD covered 1 km², encompassing sections of the Shimsha River and more distant lakes, including Maddur Lake and BV Halli Kere, located farther from the release location. Relative to the first year, the 75% UD expanded to 3 km² (a 200% increase from Year 1), while the 99% UD showed the most substantial expansion, reaching up to 86 km² (a 169% increase), reflecting increased ranging behavior and the utilization of more distant wetland networks (Figure 1).
Adult pelican (Aadi) movement patterns
Aadi was tracked for 756 days, providing 108,864 GPS locations. Immediately after release, Aadi moved away from the release site, first travelling to lakes in Bengaluru (approximately 100 km from the release location), and then to Mysuru (approximately 80 km from the release location). Throughout the tracking period, all movements remained within the Bengaluru–Mysuru geographical belt, demonstrating spatial fidelity to this corridor.
During the first non-breeding season (February to September 2023), Aadi’s movements were broadly distributed across the Bengaluru–Mysuru belt. A few concentrated 50% core areas were evident (less than 1 km²), indicating spatially diffuse foraging patterns. The 75% UD encompassed 1 km², centered on Kudlur Lake in a neighboring district, while the 99% UD extended to 907 km², incorporating multiple urban lakes in both the Bengaluru and Mysuru regions. In the second non-breeding season (May to September 2024), ranging patterns contracted substantially. The 50% core area was recorded to be 2 km², concentrated within a 10 km radius of Ranganathittu Bird Sanctuary in Mysuru and Karanji Lake. The 75% UD reduced to 1 km² (no change in absolute area but representing refined spatial concentration), and the 99% UD contracted to 152 km², showing an 83% reduction compared to the first non-breeding season (Figure 2).
During the first breeding season (October 2023 to April 2024), Aadi attempted breeding twice. The initial attempt at the release location (Kokkare Bellur) failed after one month of nesting. Following this failure, Aadi moved to Ranganathittu Bird Sanctuary and initiated a second nesting attempt, successfully producing one fledgling, as confirmed through photographic evidence by forest staff (pers. comm.). The 50% core area encompassed 8 km², including both breeding sites and
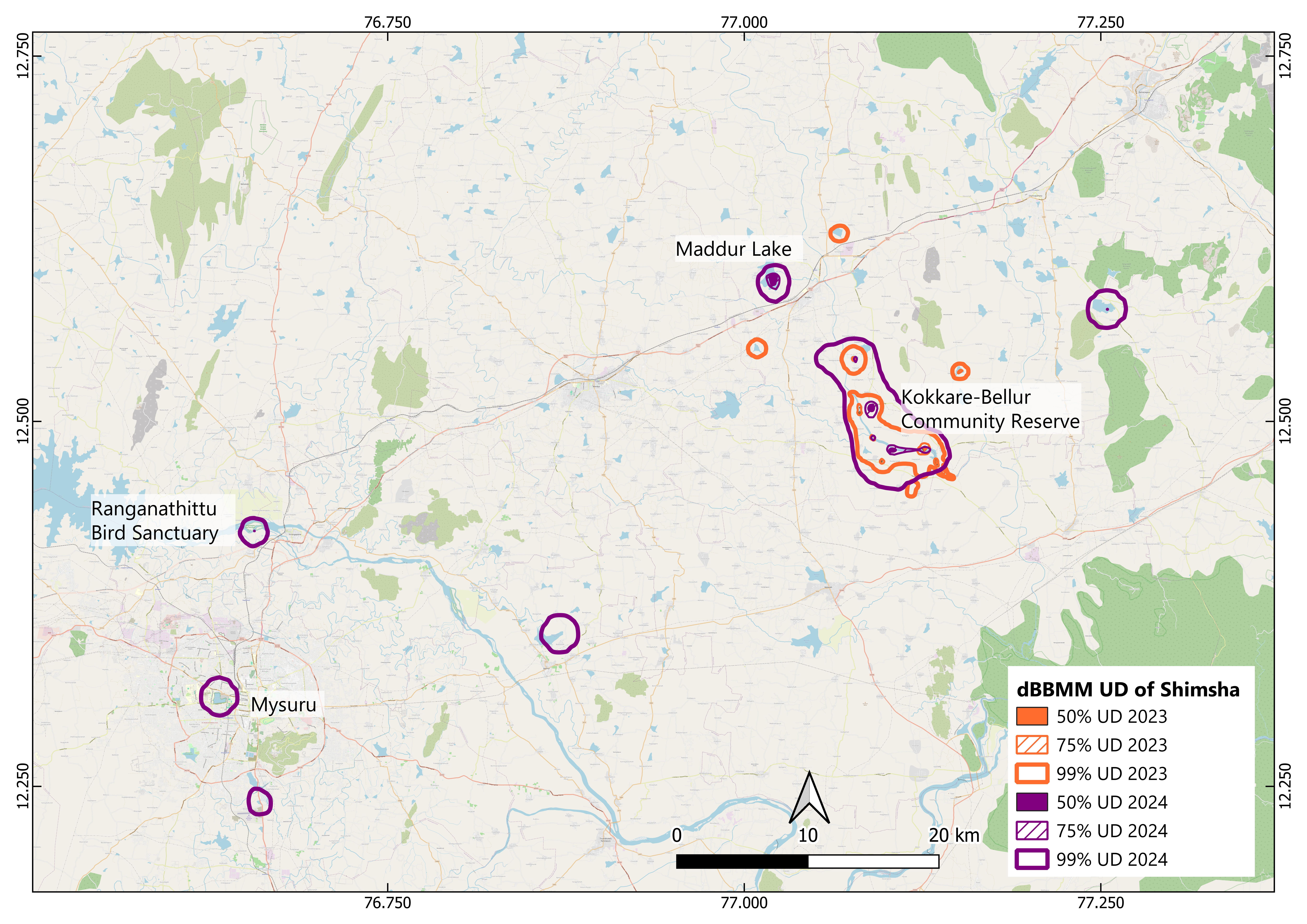
Figure 1. Utilization distributions (UD) of the juvenile Sspot-billed Ppelican (Shimsha) derived from Dynamic Brownian Bridge Movement Models (dBBMM). The maps show the 50% (core), 75% (moderate), and 99% (overall) UDs for the bird-year 2023 (November 2022–November 2023) and the bird-year 2024 (November 2023–November 2024), illustrating spatial expansion over time. Basemap data: © OSM Standard, rendered in QGIS 3.28.
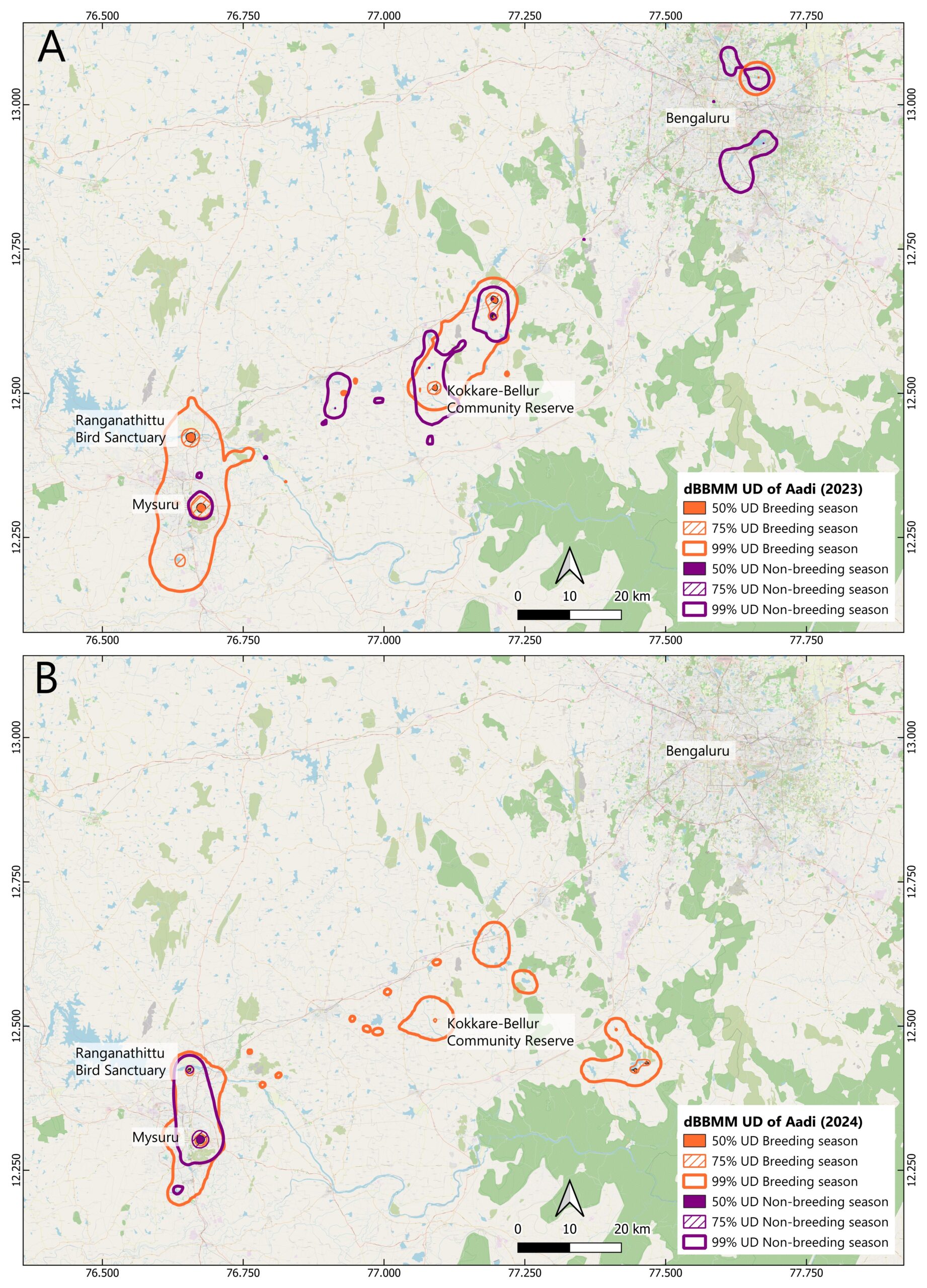
Figure 2. Inter-annual comparison of breeding and non-breeding season space use of the adult spot-billed pelican (Aadi) derived from Dynamic Brownian Bridge Movement Models (dBBMM). Panels represent (A) bird-year 2023, showing the non-breeding season (February–September 2023) followed by the breeding season (October 2023–April 2024) and (B) bird-year 2024, showing the non-breeding season (May–September 2024) followed by the breeding season (October 2024–February 2025). The maps depict the 50% (core), 75% (moderate), and 99% (overall) utilization distributions (UDs) for each season. Basemap data: © OSM Standard, rendered in QGIS 3.28.
neighboring lakes, which represented foraging grounds. The 75% UD extended to 43 km², whilst the 99% UD covered 703 km², capturing movements between failed and successful breeding attempts and associated foraging areas. In the second breeding season (October 2024 to February 2025), Aadi bred only once at Ranganathittu. The 50% core area was reduced to 2 km² (a 75% reduction), encompassing Ranganathittu, a neighboring lake, and a distant reservoir located approximately 120 km from Ranganathittu. The 75% UD contracted to 12 km² (a 72% reduction), and the 99% UD decreased to 471 km² (a 33% reduction) (Figure 2).
Daily distance travelled (DDT) differed markedly between the adult (Aadi) and juvenile (Shimsha) and varied across seasons and years. For the adult pelican, Aadi, median daily distance travelled (DDT) increased consistently during breeding seasons relative to the preceding non-breeding periods. In 2023, the median DDT during the non-breeding season was 5.55 km (range: 1.96–99.2 km), increasing to 24.7 km (range: 1.56–151 km) during the 2023 breeding season. Similarly, in 2024, the median DDT increased from 1.39 km (range: 0.41–66.7 km) in the non-breeding period to 3.54 km (range: 0.39–73.3 km) during the breeding season. The juvenile pelican, Shimsha, showed consistent daily movement patterns across years, with comparatively narrower seasonal variation. During the first- year post-release (2022–2023), the median DDT was 8.8 km, with values ranging from 1.47 km to 68.7 km. In the second year (2023–2024), the median DDT increased to 10.9 km, while the overall range remained similar (0.41–67.6 km). Compared to the adult, Shimsha displayed higher median DDT values than Aadi during non-breeding periods, but lower maximum DDT than those recorded by the adult during breeding (Figure 3).
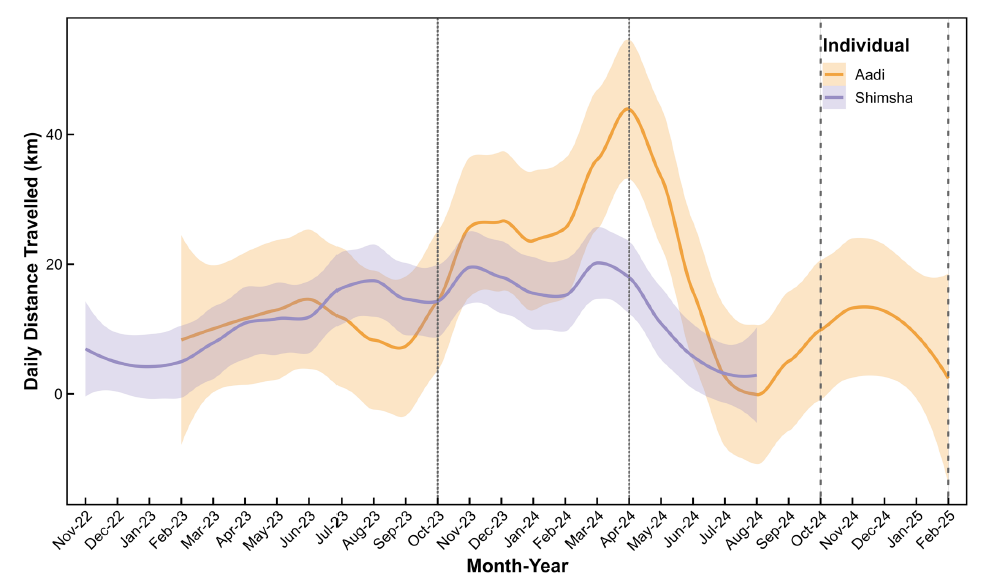
Figure 3. Comparison of age-dependent movement patterns in two spot-billed pelicans tracked using GPS telemetry. The juvenile (Shimsha) shows a progressive increase in daily distance travelled over time, whereas the adult (Aadi) exhibits a cyclical movement pattern with pronounced peaks in daily travel during breeding seasons, consistent with central-place foraging behaviour. Vertical dashed lines indicate the onset and termination of Aadi’s breeding periods (October 2023–April 2024 and October 2024–February 2025). Lines represent LOESS-smoothed mean daily distance travelled, with shaded bands indicating 95% confidence intervals.
Discussion
This study provides novel insights into the post-release movement ecology of rescued spot-billed pelicans, revealing distinct ranging strategies between a hand-raised juvenile and a rehabilitated adult. The application of GPS telemetry, coupled with dBBMM analysis, enabled the precise quantification of utilization distributions and temporal dynamics in space use, contributing to an understanding of individual-level variation in this Near Threatened species.
The movement patterns of Shimsha and Aadi reflect fundamental differences in life-history stage and prior experience. Shimsha’s gradual spatial expansion and increasing daily distances traveled align with the ontogenetic development of foraging skills and spatial knowledge acquisition in juvenile waterbirds (Mendez et al., 2017). The progressive shift from localized movements near the release site to exploration of more distant wetlands suggests increasing independence and competence in identifying foraging opportunities. Hand-raised waterbirds often exhibit protracted development of optimal foraging behaviors compared to wild-reared conspecifics (Delord et al., 2024), and the gradual increase observed here suggests Shimsha acquired foraging skills over an extended period post-release. By Year 2, daily distances approached those of the adult during non-breeding periods, indicating functional integration into wild foraging patterns.
In contrast, Aadi’s immediate long-distance departure from the release site and rapid establishment of movements within the Bengaluru–Mysuru corridor indicate retention of spatial memory and navigational abilities despite rehabilitation. This behavioral pattern suggests that adult individuals possess prior knowledge of regional wetland networks, enabling them to navigate efficiently to known foraging and breeding areas (Baert et al., 2022). The restriction of movements to this corridor throughout the tracking period demonstrates spatial fidelity, a common trait among adult waterbirds with established home ranges.
Aadi’s evident seasonal variation in space use, with contracted core areas during the second year, reflects habitat familiarity and optimized resource use. The dramatic reduction in UD size during the non-breeding season in Year 2, with movements restricted to the vicinity of Ranganathittu and Karanji Lake, suggests a refinement of foraging strategies and the identification of productive, reliable wetlands. Such spatial optimization has been documented in other colonial waterbirds, where experienced individuals concentrate their foraging effort in familiar, high-quality habitats rather than engaging in exploratory movements (Geary et al., 2020).
The breeding season patterns of Aadi reveal reproductive site fidelity and flexible nesting strategies. The dual breeding attempts in Year 1, including a failed attempt followed by successful breeding at an alternative site, demonstrate behavioral plasticity in response to initial failure. Site-switching following breeding failure is an adaptive strategy in colonial waterbirds, enabling relocation to sites with higher success probability (Danchin et al., 1998; Wilkinson & Jodice, 2022). The subsequent concentration of breeding activity at Ranganathittu in Year 2, reflected by a reduced UD size, indicates the development of site fidelity following successful reproduction, a common pattern in long-lived colonial species (Boulinier & Danchin, 1997).
The elevated daily distances traveled recorded for Aadi during breeding seasons reflect increased foraging effort associated with provisioning demands. Spot-billed pelicans are known to travel substantial distances between breeding colonies and foraging wetlands (Kannan & Manakadan, 2005), and the observed 35 to 42 km/day movements during reproduction align with central-place foraging constraints. The post-breeding decline in DDT suggests reduced energetic demands and potentially greater prey availability or accessibility during non-breeding periods.
From a conservation perspective, the identification of Ranganathittu and associated lakes as critical year-round habitats for Aadi, as well as the Shimsha River corridor, highlights the importance of protecting wetland networks rather than isolated sites. Connectivity between breeding colonies and foraging wetlands is essential for pelican persistence, particularly given the seasonal variation in water availability across India’s wetlands (Kannan & Pandiyan, 2013). These findings underscore the need for landscape-level wetland management strategies that maintain hydrological connectivity, regulate disturbance during breeding seasons, and prioritize the protection of interconnected lake systems within the Bengaluru–Mysuru corridor. Such integrated conservation planning will be critical for sustaining breeding populations of spot-billed pelicans in human-dominated landscapes.
The successful post-release survival and breeding of Aadi over two consecutive seasons demonstrates the conservation value of rehabilitation and release programs for spot-billed pelicans. The confirmed breeding success at Ranganathittu indicates that rehabilitated adults can successfully reintegrate into wild populations and contribute at the population level. Similarly, Shimsha’s apparent successful transition to independent foraging suggests that hand-raised juveniles, despite extended human exposure, can develop necessary survival skills.
This case report is limited by a small sample size (n = 2), precluding statistical generalization to broader populations. Individual variation in movement ecology can be substantial, and the rescued status of these pelicans may influence ranging behavior compared to colony-reared individuals. Future studies should track larger samples, including wild-caught individuals across multiple colonies, to establish population-level movement patterns and identify whether rescued individuals exhibit behavioral differences. Additionally, incorporating individual marking techniques such as color banding or wing tagging would facilitate long-term behavioral monitoring, site-fidelity assessments, and individual-based movement analyses. For tagged juveniles such as Shimsha, continued monitoring through sexual maturity would reveal whether juvenile spatial patterns predict adult home range establishment. Notably, wing-tagging of 14 pelicans was conducted during 2021-2023, and their resightings have been systematically monitored, providing complementary individual-level data. This study focused on spatial patterns without integrating data on habitat characteristics or prey availability. Future research should incorporate wetland productivity metrics, fish abundance data, and anthropogenic disturbance indices to model habitat selection and identify drivers of space use variation. The integration of accelerometer data could refine activity budgets and distinguish between foraging and non-foraging movements. Long-term tracking beyond two years would enable assessment of lifespan movement dynamics, breeding site fidelity over multiple seasons, and potential dispersal patterns.
Acknowledgement
The authors would like to thank the Karnataka Forest Department, particularly the Mysuru Wildlife Division, for financially supporting this research (A2/ACT/KBCR/TAG/2020-21, dated 08 March 2021). We are grateful to Lokesh P. Gowda, Manish Manick, and Manjunathan J. for their valuable assistance during the field tagging process. We thank veterinarians who supported the project, Dr. Satish P., Dr. H.S. Prayag, Dr. Roopa, Dr. Vaseem Mirza, and Dr. M. Karikalan. Our appreciation is extended to the local community of Kokkare-Bellur for their continued cooperation and support, which made this study possible. All field observations and research activities were conducted in compliance with the current laws and regulations of India.
CONFLICT OF INTEREST
R. Suresh Kumar is an Academic Editor at the Journal of Wildlife Science. However, he did not participate in the peer review process of this article except as an author. The authors declare no other conflict of interest.
DATA AVAILABILITY
Data is available on request from the corresponding author.
AUTHORS’ CONTRIBUTION
All the authors contributed to the conception and design of the study. Material prepa-ration and data collection were performed by Aksheeta Mahapatra. The analysis was performed by Aksheeta Mahapatra and Harindra L. Baraiya. The first draft of the man-uscript was written by Aksheeta Mahapatra, and all the authors reviewed, edited, and approved the final manuscript.
ETHICS STATEMENT
All procedures involving animal handling were conducted in compliance with the Wildlife (Protection) Act, 1972 of India. Permission for rescue, rehabilitation, handling, and GPS tagging of Spot-billed Pelicans was granted by the Principal Chief Conserva¬tor of Forests (PCCF), Karnataka State Forest Department. All veterinary examination, treatment, and tagging were carried out by authorized veterinarians deputed by the Karnataka Forest Department. The study did not involve invasive sampling. The re-search adhered to national wildlife welfare regulations and ethical standards for the handling of protected species, and complied with the principles of the Convention on Biological Diversity and CITES.
DECLARATION OF THE USE OF GENERATIVE AI
The authors used Gemini 2 Pro for language improvement and paraphrasing during the preparation of this manuscript. The authors thoroughly reviewed and edited all content generated using this tool and take full responsibility for the accuracy, integrity, and originality of the content of the publications.
Edited By
Ashish Jha
Wildlife Institute of India, Dehradun, India.
*CORRESPONDENCE
R Suresh Kumar
✉ suresh.wii@gmail.com
CITATION
Mahapatra, A., Baraiya, H. L. & Kumar, R. S. (2026). Post-release dispersal, space use, and breeding integration of rescued adult and juvenile spot-billed pelicans tracked using GPS telemetry. Journal of Wildlife Science, 3(1), 20-25. https://doi.org/10.63033/JWLS.CLZI7874
FUNDING
This study was supported by the Karnataka State Forest Department, Mysuru Wildlife Division, under project approval number PCCF (WL)/E2/ CR-28/2019–20.
COPYRIGHT
© 2026 Mahapatra, Baraiya & Kumar. This is an open-access article, immediately and freely available to read, download, and share. The information contained in this article is distributed under the terms of the Creative Commons Attribution License (CC BY 4.0), allowing for unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited in accordance with accepted academic practice. Copyright is retained by the author(s).
PUBLISHED BY
Wildlife Institute of India, Dehradun, 248 001 INDIA
PUBLISHER'S NOTE
The Publisher, Journal of Wildlife Science or Editors cannot be held responsible for any errors or consequences arising from the use of the information contained in this article. All claims expressed in this article are solely those of the author(s) and do not necessarily represent those of their affiliated organisations or those of the publisher, the editors and the reviewers. Any product that may be evaluated or used in this article or claim made by its manufacturer is not guaranteed or endorsed by the publisher.
Amat, J. A. & Green, A. J. (2009). Waterbirds as bioindicators of environmental conditions. In: Boon, P. J. & Raven, P. J. (eds.), Conservation Monitoring in Freshwater Habitats: A Practical Guide and Case Studies. Springer, Dordrecht. pp.45-52. https://doi.org/10.1007/978-1-4020-9278-7_5
Baert J. M., Stienen E. W. M., Verbruggen F., de Weghe N. V. Lens, L. & Müller, W. (2022). Resource predictability drives interannual variation in migratory behavior in a long-lived bird. Behavioral Ecology, 33(1), 59–68. https://doi.org/10.1093/beheco/arab132
Bernardo, C. S. S., Lloyd, H., Olmos, F., Cancian, L. F. & Galetti, M. (2011). Using post‐release monitoring data to optimize avian reintroduction programs: a 2‐year case study from the Brazilian Atlantic Rainforest. Animal Conservation, 14(6), 676–686. https://doi.org/10.1111/j.1469-1795.2011.00473.x
BirdLife International. (2017). Pelecanus philippensis. The IUCN Red List of Threatened Species 2017. https://datazone.birdlife.org/species/factsheet/spot-billed-pelican-pelecanus-philippensis (Accessed on 04 January 2026)
Boulinier T. & Danchin E. (1997). The use of conspecific reproductive success for breeding patch selection in territorial migratory species. Evolutionary Ecology, 11, 505–517. https://doi.org/10.1007/s10682-997-1507-0
Danchin E., Boulinier T. & Massot M. (1998). Conspecific reproductive success and breeding habitat selection: implications for the study of coloniality. Ecological Society of America, 79(7), 2415–2428. https://doi.org/10.1890/0012-9658(1998)079[2415:CRSABH]2.0.CO;2
del Hoyo, J., Elliott, A. & Sargatal, J. (eds.), (1992). Handbook of the Birds of the World, Volume 1: Ostrich to Ducks. Lynx Edicions, Barcelona.
Delord, K., Weimerskirch, H. & Barbraud, C. (2024). The challenges of independence: ontogeny of at-sea behaviour in a long-lived seabird. Peer Community Journal, 4, e30. https://doi.org/10.24072/pcjournal.386
Durairajan, R., Ramya, R., Jayanthi, N., Soundararajan, C., Azhahianambi, P., Murugan, M., Ramesh, J. & Sridhar, R. (2023). Tetrameres proventriculitis and renal nematodiasis infection in pelican from Melmaruvathur area of Tamil Nadu. Theoretical Biology Forum, 12(2), 60-64.
Fiorello, C. V., Jodice, P. G., Lamb, J., Satgé, Y., Mills, K. & Ziccardi, M. (2021). Postrelease survival of California brown pelicans (Pelecanus occidentalis californicus) following oiling and rehabilitation after the Refugio oil spill. Journal of Wildlife Diseases, 57(3), 590–600. https://doi.org/10.7589/JWD-D-20-00171
Geary, B., Leberg, P. L. & Purcell, K. M. (2020). Breeding Brown Pelicans Improve Foraging Performance as Energetic Needs Rise. Scientific Reports, 10, 1686. https://doi.org/10.1038/s41598-020-58528-z
Hernandez, S. M. (2019). Postrehabilitation release monitoring of wildlife. In: Hernandez, S. M., Barron, H. W., Miller, E. A., Aguilar, R. F. & Yabsley, M. J. (eds.), Medical Management of Wildlife Species: A Guide for Practitioners. Wiley-Blackwell, Hoboken. pp.123-127. https://doi.org/10.1002/9781119036708.ch10
Hijmans, R. J., van Etten, J., Sumner, M., Cheng, J., Baston, D., Bevan, A., Bivand, R., Busetto, L., Canty, M., et al. (2025). raster: Geographic Data Analysis and Modeling. Version 3.6-32. R package. https://doi.org/10.32614/CRAN.package.raster (Accessed on 02 January 2026)
Kannan, R. & Pandiyan, J. (2013). A review on the Spot-billed Pelican Pelecanus philippensis literature. Frontiers in Biology, 8, 333–352. https://doi.org/10.1007/s11515-013-1252-4
Kannan, V. & Manakadan, R. (2005). The status and distribution of Spot-billed Pelican Pelecanus philippensis in southern India. Forktail, 21, 9–14.
Kranstauber, B., Smolla, M. & Scharf, A. K. (2024). move: Visualizing and Analyzing Animal Track Data. Version 4.2.6. R package. https://doi.org/10.32614/CRAN.package.move (Accessed on 02 January 2026)
Kranstauber B., Kays R., LaPoint S. D., Wikelski M. & Safi K. (2012). A dynamic Brownian bridge movement model to estimate utilization distributions for heterogeneous animal movement. Journal of Animal Ecology, 81(4), 738–746. https://doi.org/10.1111/j.1365-2656.2012.01955.x
Kumar, S., Periyasamy, A., Rao, N. V. R., Sunil, S. S., Kumara, H. N., Sundararaj, P., Chidananda, G. & Sathish, A. (2019). Multiple infestations of gastrointestinal parasites – probable cause of high mortality of Spot-billed Pelican (Pelecanus philippensis) at Kokrebellur Community Reserve, India. International Journal for Parasitology: Parasites and Wildlife, 9, 68–73. https://doi.org/10.1016/j.ijppaw.2019.04.001
Lamb, J. S., Fiorello, C. V., Satgé, Y. G., Mills, K., Ziccardi, M. & Jodice, P. G. (2018). Movement patterns of California brown pelicans (Pelecanus occidentalis californicus) following oiling and rehabilitation. Marine Pollution Bulletin, 131(A), 22–31. https://doi.org/10.1016/j.marpolbul.2018.03.043
Mathews, A., Malathi, S., Mohinikumari, P. & Shameem, U. (2025). Report on nematode parasites in Spot-billed Pelicans (Pelecanus philippensis) and Painted Storks (Mycteria leucocephala) from Telineelapuram, India. Journal of Parasitic Diseases, 1–19. https://doi.org/10.1007/s12639-025-01842-y
Mendez, L., Prudor, A. & Weimerskirch, H. (2017). Ontogeny of foraging behaviour in juvenile Red-footed Boobies (Sula sula). Scientific Reports, 7, 13886. https://doi.org/10.1038/s41598-017-14478-7
Pebesma, E., Bivand, R., Rowlingson, B., Gomez-Rubio, V., Hijmans, R., Sumner, M., MacQueen, D., Lemon, J., Lindgren, F., O’Brien, J., O’Rourke, J. & Hausmann, P. (2025a). sp: Classes and Methods for Spatial Data. Version 2.2-0. R package. https://doi.org/10.32614/CRAN.package.sp (Accessed on 02 January 2026)
Pebesma, E., Bivand, R., Racine, E., Sumner, M., Cook, I., Keitt, T., Lovelace, R., Wickham, H., Ooms, J., Müller, K., Pedersen, T. L., Baston, D. & Dunnington, D. (2025b). sf: Simple Features for R. Version 1.0-24. R package. https://doi.org/10.32614/CRAN.package.sf (Accessed on 02 January 2026)
Pyke, G. H. & Szabo, J. K. (2018). Conservation and the 4 Rs, which are rescue, rehabilitation, release, and research. Conservation. Biology, 32, 50–59. https://doi.org/10.1111/cobi.12937
R Core Team. (2024). R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/ (Accessed on 02 January 2026)
Raine, A. F., Anderson, T., Vynne, M., Driskill, S., Raine, H. & Adams, J. (2020). Post-release survival of fallout Newell’s Shearwater fledglings from a rescue and rehabilitation program on Kaua‘i, Hawai‘i. Endangered Species Research, 43, 39–50. https://doi.org/10.3354/esr01051
Rozsypalová, L., Literák, I., Raab, R., Peške, L., Krone, O., Škrábal, J. & Meyburg, B. U. (2025). Survival of White-tailed Eagles tracked after rehabilitation and release. Journal of Raptor Research, 59(1), 1–16. https://doi.org/10.3356/jrr2417
Wilkinson, B. P. & Jodice, P. G. R. (2022). Interannual colony exchange among breeding Eastern Brown Pelicans. Journal of Field Ornithology, 93(1), 5. https://doi.org/10.5751/JFO-00074-930105
Edited By
Ashish Jha
Wildlife Institute of India, Dehradun, India.
*CORRESPONDENCE
R Suresh Kumar
✉ suresh.wii@gmail.com
CITATION
Mahapatra, A., Baraiya, H. L. & Kumar, R. S. (2026). Post-release dispersal, space use, and breeding integration of rescued adult and juvenile spot-billed pelicans tracked using GPS telemetry. Journal of Wildlife Science, 3(1), 20-25. https://doi.org/10.63033/JWLS.CLZI7874
FUNDING
This study was supported by the Karnataka State Forest Department, Mysuru Wildlife Division, under project approval number PCCF (WL)/E2/ CR-28/2019–20.
COPYRIGHT
© 2026 Mahapatra, Baraiya & Kumar. This is an open-access article, immediately and freely available to read, download, and share. The information contained in this article is distributed under the terms of the Creative Commons Attribution License (CC BY 4.0), allowing for unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited in accordance with accepted academic practice. Copyright is retained by the author(s).
PUBLISHED BY
Wildlife Institute of India, Dehradun, 248 001 INDIA
PUBLISHER'S NOTE
The Publisher, Journal of Wildlife Science or Editors cannot be held responsible for any errors or consequences arising from the use of the information contained in this article. All claims expressed in this article are solely those of the author(s) and do not necessarily represent those of their affiliated organisations or those of the publisher, the editors and the reviewers. Any product that may be evaluated or used in this article or claim made by its manufacturer is not guaranteed or endorsed by the publisher.
Amat, J. A. & Green, A. J. (2009). Waterbirds as bioindicators of environmental conditions. In: Boon, P. J. & Raven, P. J. (eds.), Conservation Monitoring in Freshwater Habitats: A Practical Guide and Case Studies. Springer, Dordrecht. pp.45-52. https://doi.org/10.1007/978-1-4020-9278-7_5
Baert J. M., Stienen E. W. M., Verbruggen F., de Weghe N. V. Lens, L. & Müller, W. (2022). Resource predictability drives interannual variation in migratory behavior in a long-lived bird. Behavioral Ecology, 33(1), 59–68. https://doi.org/10.1093/beheco/arab132
Bernardo, C. S. S., Lloyd, H., Olmos, F., Cancian, L. F. & Galetti, M. (2011). Using post‐release monitoring data to optimize avian reintroduction programs: a 2‐year case study from the Brazilian Atlantic Rainforest. Animal Conservation, 14(6), 676–686. https://doi.org/10.1111/j.1469-1795.2011.00473.x
BirdLife International. (2017). Pelecanus philippensis. The IUCN Red List of Threatened Species 2017. https://datazone.birdlife.org/species/factsheet/spot-billed-pelican-pelecanus-philippensis (Accessed on 04 January 2026)
Boulinier T. & Danchin E. (1997). The use of conspecific reproductive success for breeding patch selection in territorial migratory species. Evolutionary Ecology, 11, 505–517. https://doi.org/10.1007/s10682-997-1507-0
Danchin E., Boulinier T. & Massot M. (1998). Conspecific reproductive success and breeding habitat selection: implications for the study of coloniality. Ecological Society of America, 79(7), 2415–2428. https://doi.org/10.1890/0012-9658(1998)079[2415:CRSABH]2.0.CO;2
del Hoyo, J., Elliott, A. & Sargatal, J. (eds.), (1992). Handbook of the Birds of the World, Volume 1: Ostrich to Ducks. Lynx Edicions, Barcelona.
Delord, K., Weimerskirch, H. & Barbraud, C. (2024). The challenges of independence: ontogeny of at-sea behaviour in a long-lived seabird. Peer Community Journal, 4, e30. https://doi.org/10.24072/pcjournal.386
Durairajan, R., Ramya, R., Jayanthi, N., Soundararajan, C., Azhahianambi, P., Murugan, M., Ramesh, J. & Sridhar, R. (2023). Tetrameres proventriculitis and renal nematodiasis infection in pelican from Melmaruvathur area of Tamil Nadu. Theoretical Biology Forum, 12(2), 60-64.
Fiorello, C. V., Jodice, P. G., Lamb, J., Satgé, Y., Mills, K. & Ziccardi, M. (2021). Postrelease survival of California brown pelicans (Pelecanus occidentalis californicus) following oiling and rehabilitation after the Refugio oil spill. Journal of Wildlife Diseases, 57(3), 590–600. https://doi.org/10.7589/JWD-D-20-00171
Geary, B., Leberg, P. L. & Purcell, K. M. (2020). Breeding Brown Pelicans Improve Foraging Performance as Energetic Needs Rise. Scientific Reports, 10, 1686. https://doi.org/10.1038/s41598-020-58528-z
Hernandez, S. M. (2019). Postrehabilitation release monitoring of wildlife. In: Hernandez, S. M., Barron, H. W., Miller, E. A., Aguilar, R. F. & Yabsley, M. J. (eds.), Medical Management of Wildlife Species: A Guide for Practitioners. Wiley-Blackwell, Hoboken. pp.123-127. https://doi.org/10.1002/9781119036708.ch10
Hijmans, R. J., van Etten, J., Sumner, M., Cheng, J., Baston, D., Bevan, A., Bivand, R., Busetto, L., Canty, M., et al. (2025). raster: Geographic Data Analysis and Modeling. Version 3.6-32. R package. https://doi.org/10.32614/CRAN.package.raster (Accessed on 02 January 2026)
Kannan, R. & Pandiyan, J. (2013). A review on the Spot-billed Pelican Pelecanus philippensis literature. Frontiers in Biology, 8, 333–352. https://doi.org/10.1007/s11515-013-1252-4
Kannan, V. & Manakadan, R. (2005). The status and distribution of Spot-billed Pelican Pelecanus philippensis in southern India. Forktail, 21, 9–14.
Kranstauber, B., Smolla, M. & Scharf, A. K. (2024). move: Visualizing and Analyzing Animal Track Data. Version 4.2.6. R package. https://doi.org/10.32614/CRAN.package.move (Accessed on 02 January 2026)
Kranstauber B., Kays R., LaPoint S. D., Wikelski M. & Safi K. (2012). A dynamic Brownian bridge movement model to estimate utilization distributions for heterogeneous animal movement. Journal of Animal Ecology, 81(4), 738–746. https://doi.org/10.1111/j.1365-2656.2012.01955.x
Kumar, S., Periyasamy, A., Rao, N. V. R., Sunil, S. S., Kumara, H. N., Sundararaj, P., Chidananda, G. & Sathish, A. (2019). Multiple infestations of gastrointestinal parasites – probable cause of high mortality of Spot-billed Pelican (Pelecanus philippensis) at Kokrebellur Community Reserve, India. International Journal for Parasitology: Parasites and Wildlife, 9, 68–73. https://doi.org/10.1016/j.ijppaw.2019.04.001
Lamb, J. S., Fiorello, C. V., Satgé, Y. G., Mills, K., Ziccardi, M. & Jodice, P. G. (2018). Movement patterns of California brown pelicans (Pelecanus occidentalis californicus) following oiling and rehabilitation. Marine Pollution Bulletin, 131(A), 22–31. https://doi.org/10.1016/j.marpolbul.2018.03.043
Mathews, A., Malathi, S., Mohinikumari, P. & Shameem, U. (2025). Report on nematode parasites in Spot-billed Pelicans (Pelecanus philippensis) and Painted Storks (Mycteria leucocephala) from Telineelapuram, India. Journal of Parasitic Diseases, 1–19. https://doi.org/10.1007/s12639-025-01842-y
Mendez, L., Prudor, A. & Weimerskirch, H. (2017). Ontogeny of foraging behaviour in juvenile Red-footed Boobies (Sula sula). Scientific Reports, 7, 13886. https://doi.org/10.1038/s41598-017-14478-7
Pebesma, E., Bivand, R., Rowlingson, B., Gomez-Rubio, V., Hijmans, R., Sumner, M., MacQueen, D., Lemon, J., Lindgren, F., O’Brien, J., O’Rourke, J. & Hausmann, P. (2025a). sp: Classes and Methods for Spatial Data. Version 2.2-0. R package. https://doi.org/10.32614/CRAN.package.sp (Accessed on 02 January 2026)
Pebesma, E., Bivand, R., Racine, E., Sumner, M., Cook, I., Keitt, T., Lovelace, R., Wickham, H., Ooms, J., Müller, K., Pedersen, T. L., Baston, D. & Dunnington, D. (2025b). sf: Simple Features for R. Version 1.0-24. R package. https://doi.org/10.32614/CRAN.package.sf (Accessed on 02 January 2026)
Pyke, G. H. & Szabo, J. K. (2018). Conservation and the 4 Rs, which are rescue, rehabilitation, release, and research. Conservation. Biology, 32, 50–59. https://doi.org/10.1111/cobi.12937
R Core Team. (2024). R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/ (Accessed on 02 January 2026)
Raine, A. F., Anderson, T., Vynne, M., Driskill, S., Raine, H. & Adams, J. (2020). Post-release survival of fallout Newell’s Shearwater fledglings from a rescue and rehabilitation program on Kaua‘i, Hawai‘i. Endangered Species Research, 43, 39–50. https://doi.org/10.3354/esr01051
Rozsypalová, L., Literák, I., Raab, R., Peške, L., Krone, O., Škrábal, J. & Meyburg, B. U. (2025). Survival of White-tailed Eagles tracked after rehabilitation and release. Journal of Raptor Research, 59(1), 1–16. https://doi.org/10.3356/jrr2417
Wilkinson, B. P. & Jodice, P. G. R. (2022). Interannual colony exchange among breeding Eastern Brown Pelicans. Journal of Field Ornithology, 93(1), 5. https://doi.org/10.5751/JFO-00074-930105




