TYPE: Natural History Note![]()
Barn Swallows of the Imphal Valley – a potential case of past climatic events leading to year-round residency in the population in Northeast India
RECEIVED 14 January 2026
ACCEPTED 13 February 2026
ONLINE EARLY 25 March 2026
PUBLISHED 30 March 2026
Abstract
Birds have shown notable expansions and contractions of breeding ranges in response to climate change, land-use transformation, and the creation of novel human-made habitats. Synanthropic species, those closely associated with humans, are often at the forefront of such shifts. The Barn swallow (Hirundo rustica) is a widespread synanthrope that has successfully expanded its range across most continents. In India, two subspecies of barn swallows are reported to breed in the Himalaya, while one is believed to winter in parts of Northeast India. To ascertain their occurrence range, we conducted surveys in 2022 and 2023 in Manipur and recorded the presence of a resident breeding population of Barn Swallow for the first time. We captured 45 individuals and examined their plumage variation and morphometric characteristics. Mean wing length ranged from 111–123 mm in males and 111–122 mm in females. Comparisons with available museum records of other Indian subspecies showed overlapping morphometric measurements, preventing unambiguous subspecies-level identification. This record extends the known breeding distribution of the Barn Swallow in India and highlights unresolved questions regarding subspecies limits and breeding range dynamics in the eastern part of the subcontinent.
Keywords: Breeding range, colonization, hybridization, plumage variation, subspecies limits.
Introduction
What drives the occurrence of a species remains a fundamental question in natural history. Many argue that the species occurrence is the result of the combined influence of ecological and historical processes that shape niche availability, dispersal, and colonization (Parmesan et al., 2005; Gaston & Fuller, 2009; Bridle & Hoffmann, 2022). But the global environment and anthropogenic changes, including climate, land-use modifications, add to this complexity sometimes leading to biotic homogenization. Owing to these changes, many species of birds have shown prominent shifts, both expansion and contraction, in their breeding ranges (Massimino et al., 2015; Pigot et al., 2010; Rodríguez, 2002). Interestingly, species that benefit by associating with humans – the commensals – exploit novel and widespread anthropogenic habitats and thereby show noticeable shifts in their occurrence range (Fawthrop et al., 2025).
The barn swallow (Hirundo rustica) is one such human-commensal that is well known among the local people across its distribution range owing to its nature of building its cup-shaped mud nest in human-inhabited areas. As a consequence, it is the most widespread and abundant swallow species in the world, breeding in Eurasia and North America and migrating long-distances during winter to the global south; typically, from tropical Africa, northern Australia, to Central and South America (Turner, 2006). Large distributional range has resulted in different breeding populations, and phylogeographic studies have hypothesized the role of glacial-interglacial periods in shaping the population divergence primarily due to range expansions and contractions in response to climatic shifts (Turner & Rose, 2010).
Across the globe, six subspecies of barn swallows have been recognized, of which four are strict migrants, and two are residents (Dor et al., 2010; Turner & Rose, 2010). The nominate, H. r. rustica (Europe, North Africa, and Western Asia), is the largest in body size, with a white to pale-buff vent and a moderately broad, complete breast band. The Asian subspecies, H. r. gutturalis, is small, buff-bellied, with a narrower and broken breast band. The Siberian subspecies, H. r. tytleri, is intermediate in body size, with a narrow, complete breast band and dark brown vent (Scordato & Safran, 2014). The American subspecies, H. r. erythrogaster, is similar to H. r. gutturalis with an extensively rufous throat, broken breast band, and rufous chestnut to orange underparts. There are two sedentary subspecies of barn swallows- H. r. savignii occurs in Egypt and has a smaller body size with rufous-chestnut underparts, while the Middle Eastern subspecies, H. r. transitiva, is similar in size to H. r. rustica but has a broader breast band and much darker rufous-buff vent (Scordato et al., 2017). Except for H. r. savignii and H. r. transitiva, all others are migratory, occupying much of the southern hemisphere during the non-breeding season (Hobson et al., 2015; Liechti et al., 2015; Turner, 2006). Although subspecies vary in morphological traits (Figure 1, Dor et al., 2010; Turner & Rose, 2010; Brown & Brown, 2020), considerable overlap in these characters often complicates subspecies identification, especially in contact zones and poorly sampled regions.
In the Indian subcontinent, three subspecies of Barn Swallow have been reported by Ali & Ripley, (1987), where two – H. r. rustica and H. r. gutturalis – are believed to breed in the Himalayan region. The nominate, H. r. rustica, is thought to breed from Baluchistan till Kathmandu valley, Nepal, while H. r. gutturalis is assumed to mix with rustica in Nepal and extend its breeding eastward to North Bengal, Sikkim, Bhutan, and Arunachal Pradesh in India. The subspecies H. r. gutturalis is also reported to breed in the Assam valley, south of the River Brahmaputra. While the third, H. r. tytleri, is considered a regular winter visitor to Northeast India (Ali & Ripley, 1987). Despite these assertions, the precise breeding limits of these subspecies in India remain speculative, largely owing to the lack of any ecological study on the species in the region.
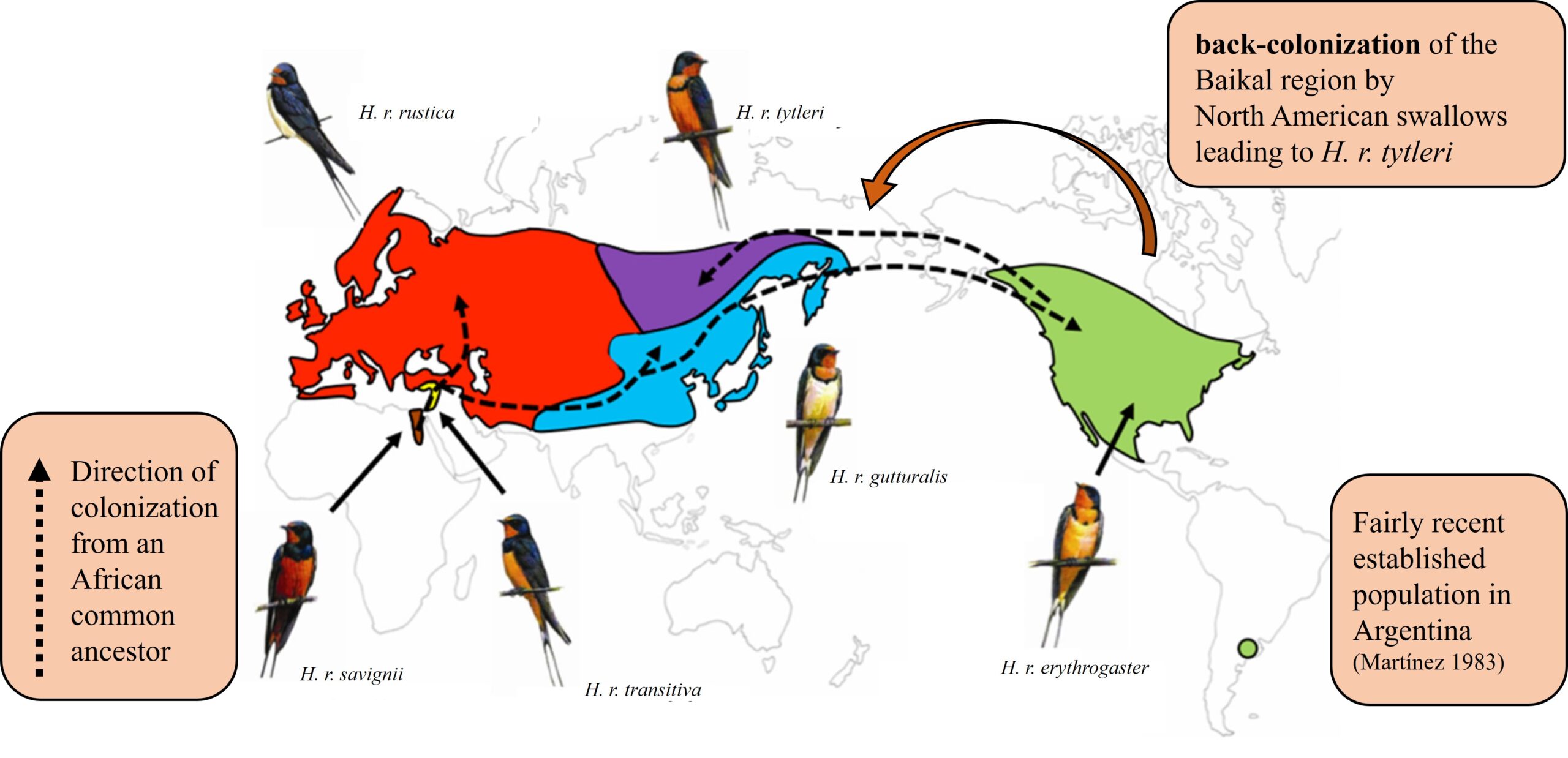
Figure 1. Range map of the six recognized subspecies of barn swallow depicting overall plumage variations in subspecies – color of underparts, breast band, and throat color. Dashed arrows show hypothesized directions of colonization of subspecies across the breeding distribution. The figure also depicts back-colonization by North American swallows (H. r. erythrogaster) to the Baikal region, which eventually separated as H. r. tytleri. (Figure adapted from Dor et al. (2010) and Scordato & Safran (2014). Barn Swallow illustrations by Hillary Burn.)
We, therefore, conducted sampling in the northeastern state of Manipur, which lies at the eastern extremity of the presumed occurrence range of the barn swallow in India. This region is geographically separated from the main Himalayan breeding areas and remains poorly studied, making it an important location for evaluating range limits and potential population differentiation. If breeding occurs in Manipur, the population may represent a peripheral or relatively isolated group compared to Himalayan breeding populations (presumably H. r. gutturalis). To evaluate the subspecies identity, we applied the traditional method of variations in morphometric traits (e.g., wing length, tail streamer length) and plumage coloration. Specifically, we compared field-based morphometric measurements with published museum data from recognized subspecies to evaluate whether the Manipur population aligns with any of the existing subspecies descriptions or overlaps with multiple subspecies.
Study area and methods
This study was conducted in the Manipur state of Northeast India, between 2022 and 2023. Rapid surveys across the state confirmed the absence of barn swallows in the hilly region, and therefore, the subsequent surveys were carried out in the Imphal Valley, a low-lying basin (~750 m asl) in the center of Manipur. Imphal is an intermontane valley, spanning an area of approximately 1864.44 km2, geographically located between 24°–25°N latitudes and 93.50°–94.25°E longitudes. The formation of this almost oval-shaped valley is a result of numerous small rivers, such as the Imphal River and its tributaries Iril, Thoubal, Khuga, and Sekmai, originating from the surrounding hills. The Imphal Valley largely falls within a subtropical monsoon‐type (Köppen: Cwa) climate (Beck et al., 2023), and experiences maximum summer temperatures ranging from 32 to 34°C, while winter temperatures can drop to a minimum of 1–2°C.
We conducted surveys radiating from the center of the valley towards its periphery, covering the districts of Imphal East, Imphal West, Bishnupur, and Thoubal. We consulted local people in locating the nests in small towns and villages, and recorded the presence of barn swallow nests associated with human structures and assessed breeding activity. During our surveys in 2023, we located barn swallow nests in approximately 50 properties, all confined to the central valley (Figure 2). At selected sites, we captured barn swallows using mist-nets and documented their plumage characteristics. We recorded standardized morphometric measurements, including body weight (in g), and length (in mm) of tarsus, head, bill, wing, and outer and inner tail length (Table 1). In discussions with local people of the valley, the Meitei, we were informed of the cultural significance of barn swallows. The Meitei community, a Tibeto-Burman ethnic group native to Manipur that largely follows the Hindu belief system (Devi & Devi, 2025), regards
Table 1. Morphometric measurements of adult barn swallows captured in Imphal Valley, Manipur. Note: Tail streamer is the difference between outer and inner tail length and is a sexually selected trait with males having longer tail streamers than females.
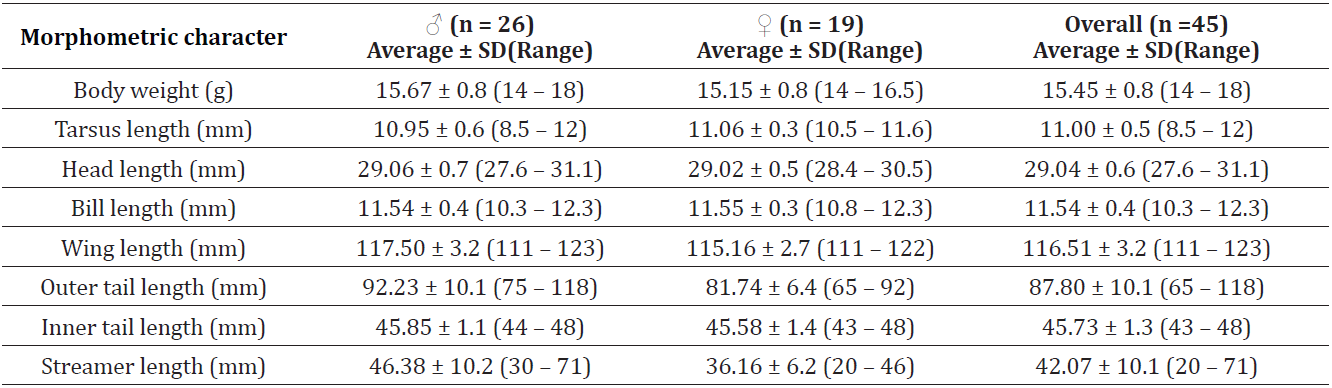
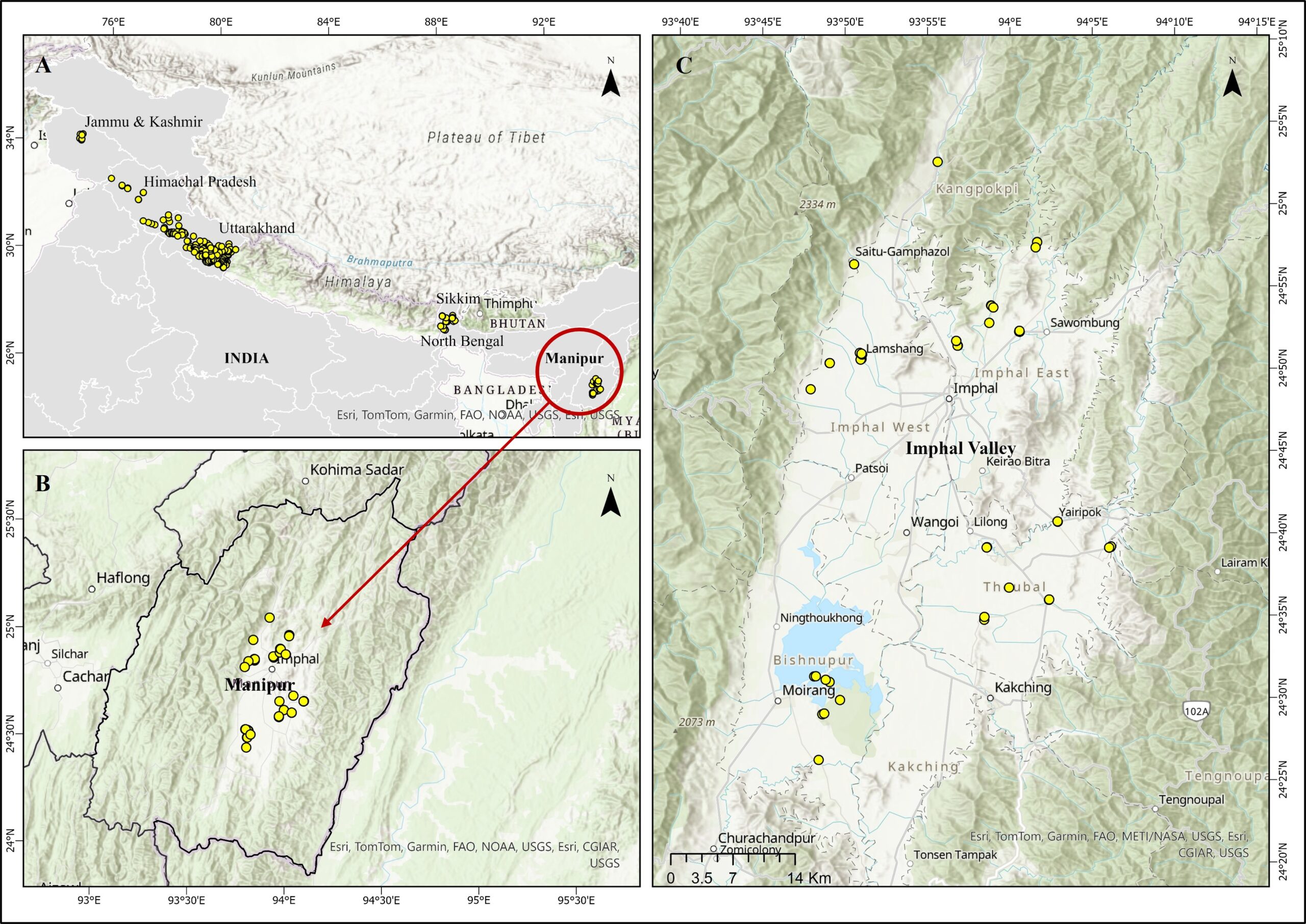
Figure 2. Distribution of barn swallow nest locations showing (A) breeding sites across the Himalayan region, (B) records from Manipur, and (C) a magnified view of nest locations within the Imphal Valley.
the barn swallow as a symbol of Goddess Lakshmi, prosperity, and good fortune. This belief translates into strong protection of nests and barn swallows within private properties, likely contributing to the persistence of breeding populations in densely inhabited areas in Imphal Valley.
Results and Discussion
During our surveys, we recorded the presence of a resident breeding population of barn swallows in Imphal Valley, reported for the first time in science. The population in Manipur was identified as year-round resident based on the continued presence of adults at nesting sites during both the breeding and non-breeding seasons observed during our field surveys, as well as information obtained from property owners where the swallows were nesting. This was further supported by our efforts of ringing adults with uniquely coded metal bands, which allowed us to confirm site fidelity over time. We captured a total of 45 adult barn swallows (26 males and 19 females) from the nests during the non-breeding season. Sex was determined using morphometric measurements, wherein males had longer tail streamers and bright colored plumage compared to females. Individuals captured in the Imphal Valley exhibited quite varying plumage color overall. The swallows were smaller-bodied, with an extensively rufous throat, with some having a broad and complete breast band, while some had a broken breast band, and rufous-chestnut to orange underparts extending to the vent (Figure 3).
The average body size (represented by wing length) was found to be 117.50 ± 3.2 mm (range: 111-123 mm) for males and 115.16 ± 2.7 mm (range: 111-122 mm) for females. The averaged outer tail length was found to be 92.23 ± 10.1 mm (range: 75-118 mm) for males, and 81.74 ± 6.4 mm (65-92 mm) for females. When comparing with the measurements of museum specimens of H. r. rustica, H. r. gutturalis and H. r. tytleri (Ali & Ripley, 1987), the measurements from Manipur’s breeding population overlapped with all three subspecies preventing unambiguous subspecies identification (Table 2). Although plumage coloration was not quantitatively assessed, qualitative observations suggested that individuals from Manipur, particularly in throat and underpart coloration, resemble the American subspecies H. r. erythrogaster and the Siberian subspecies H. r. tytleri. However, whether the Manipur population represents an intermediate form between these subspecies can also not be determined, due to substantial overlap in morphological traits and the absence of quantitative plumage analyses. These preliminary observations highlight the need for more comprehensive studies on subspecies distributions in South and Southeast Asia, which may reveal a more complex biogeographic pattern than currently recognized.
Interestingly, populations from different subspecies are also known to intermix and form hybrid zones (Scordato et al., 2017, 2020), exhibiting diverse migratory strategies. Birds from the rustica – gutturalis hybrid zone in western China migrate either westward to Africa or south across the Tibetan Plateau into India (Turbek et al., 2022), and populations breeding in the Amur contact zone winter in Southeast Asia (Heim et al., 2020). Yet uncertainties remain, particularly in Asia, where subspecies and hybrid populations exhibit diverse and poorly resolved migratory strategies. For instance, migration of the Siberian H. r. tytleri is poorly known, with only one geolocator-tracked individual suggesting a direct route into Southeast Asia (Anisimova et al., 2025). These contrasting patterns highlight significant knowledge gaps in the migratory connectivity of Asian barn swallow populations, particularly those moving into or wintering within the Indian subcontinent.
Barn swallows are known for their exceptional dispersal ability and for repeatedly colonizing new regions through both natural movements and human-mediated habitat expansion (Hobson et al., 2015; Scordato et al., 2017). Phylogenetic studies indicate that the H. rustica complex is monophyletic and structured into two major clades: a Europe–Middle East clade and an Asia–America clade (Zink et al., 2006; Dor et al., 2010). European and West Asian populations differentiated around the onset of the Holocene (Smith et al., 2018). The East and South Asian lineages colonized independently during the late Pleistocene, while the colonization of the Americas, a relatively recent event, likely occurred at least 50 thousand years ago (kya; Lombardo et al., 2022).
Notably, a secondary dispersal event from North America back into Asia at approximately 27 kya has been proposed (Figure 1), explaining the close genetic affinity between H. r. tytleri and
Table 2. Morphometric measurement of traits – wing length and outer tail length (in mm) reported by Ali & Ripley (1987) of museum specimens of H. r. rustica, H. r. gutturalis and H. r. tytleri compared with barn swallows sampled in the present study from Imphal Valley, Manipur. Sample sizes are given wherever available as (n).

H. r. erythrogaster (Zink et al. 2006). Against this backdrop, we propose two non-mutually exclusive hypotheses to explain the origin of the Manipur population. First, a secondary colonization and admixture hypothesis – where the breeding population in Manipur may have originated from H. r. tytleri, with historical introgression from H. r. erythrogaster following back-colonization into Asia, resulting in the observed intermediate phenotypic traits. This can be tested via genome-wide analyses to look for admixture signatures, as well as broader sampling across putative contact zones in Central and East Asia.
Second, a shift in migratory strategy hypothesis – the wintering H. r. tytleri populations in Northeast India may have reduced or abandoned long-distance migration and become sedentary. Similar shifts have been documented in North American barn swallows (Winkler et al., 2017), which began breeding within their former wintering range in South America during the 19th century. This scenario can be tested using tracking data (e.g., geolocators or stable isotope analyses) to determine migratory connectivity, along with long-term demographic monitoring to assess residency and recruitment patterns.
In addition, the establishment of breeding populations in the Imphal Valley may have been facilitated by favorable climatic conditions and the availability of nesting sites; evaluating this possibility would require habitat suitability modelling and comparative ecological data from adjacent regions. If supported, either scenario would suggest a notable shift in range limits and/or migratory behavior, highlighting the ecological flexibility of the barn swallow. Furthermore, whether the Manipur breeding population is demographically or genetically connected to migratory H. r. tytleri, as suggested by Ali & Ripley (1987), remains unresolved and warrants further investigation.
Significance and future directions
This study provides the first documentation of a resident breeding population of barn swallows in Manipur, extending the known breeding range of the species in the Indian subcontinent. It highlights a critical ecological gap in our understanding of avian biogeography in this under-studied region and challenges long-standing assumptions about subspecies limits. Future work should integrate detailed breeding ecology, long-term monitoring, and climate niche modelling to evaluate the drivers of residency and range expansion. Comparative studies between resident populations in Manipur and migratory Himalayan populations could yield valuable insights into life-history flexibility under changing climatic regimes. More importantly, genomic analyses will be essential to resolve the subspecies identification, their evolutionary origins and divergence times, and to test hypotheses of admixture and secondary colonization. More broadly, our findings emphasize the importance of natural history observations, particularly in culturally diverse landscapes where human attitudes can profoundly shape the distribution and persistence of synanthropic species.

Figure 3. Variation in plumage coloration in adult barn swallows sampled from the Imphal Valley, Manipur. Row A shows males with long tail streamers, and Row B shows females with shorter tail streamers. Underparts range from buff to rufous-chestnut to orange in both sexes (Row A & B). Variation is also evident in breast-band and throat characteristics (Row C), including narrow and broken breast bands (typical of H. r. gutturalis), narrow and complete breast bands (typical of H. r. tytleri), and broken breast bands diffused with rufous throat coloration (typical of H. r. erythrogaster).
Acknowledgement
The authors also thank the Wildlife Institute of India for institutional support and the Manipur Forest Department for granting the necessary permission. The authors acknowledge the support of the forest department staff of amenglong forest department who helped in carrying out surveys. DST-Inspire Fellowship Program for providing PhD fellowship to AK. The authors are also thankful to the people of Manipur who allowed the team access to their properties and capture the birds. The authors are grateful to the local field support provided by Indra.
CONFLICT OF INTEREST
R. Suresh Kumar is an Academic Editor at the Journal of Wildlife Science. However, he did not participate in the peer review process of this article except as an author. The authors declare no other conflict of interest.
DATA AVAILABILITY
Data is available on request from the corresponding author.
AUTHORS’ CONTRIBUTION
AK and RSK conceived the idea. AK collected and analysed the data and wrote the first draft of the paper. Both AK and RSK revised the initial draft and approved the final draft of the manuscript for submission.
Edited By
Gopi G.V.
Wildlife Institute of India, Dehradun, India.
*CORRESPONDENCE
R Suresh Kumar
✉ suresh.wii@gmail.com
CITATION
Kaur, A. & Kumar, R. S. (2026). Barn Swallows of the Imphal Valley – a potential case of past climatic events leading to year-round residency in the population in Northeast India. Journal of Wildlife Science, 3(1), 35-40. https://doi.org/10.63033/JWLS.GXZU3002
FUNDING
This research was supported by the PhD Fellowship awarded to Amarjeet Kaur under the DST-INSPIRE scheme of the Government of India (No. DST/INSPIRE Fellowship/2018/IF180199).
COPYRIGHT
© 2026 Kaur & Kumar. This is an open-access article, immediately and freely available to read, download, and share. The information contained in this article is distributed under the terms of the Creative Commons Attribution License (CC BY 4.0), allowing for unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited in accordance with accepted academic practice. Copyright is retained by the author(s).
PUBLISHED BY
Wildlife Institute of India, Dehradun, 248 001 INDIA
PUBLISHER'S NOTE
The Publisher, Journal of Wildlife Science or Editors cannot be held responsible for any errors or consequences arising from the use of the information contained in this article. All claims expressed in this article are solely those of the author(s) and do not necessarily represent those of their affiliated organisations or those of the publisher, the editors and the reviewers. Any product that may be evaluated or used in this article or claim made by its manufacturer is not guaranteed or endorsed by the publisher.
Ali, S. & Ripley, S. D. (1987). Handbook of the Birds of India and Pakistan. Volume 5 Larks to the Grey Hypocolius. Oxford University Press.
Anisimova, V., Anisimov, Y., Bastardot, M., Beermann, I., Kunz, F. & Heim, W. (2026). Geolocator tracking and ring recoveries reveal the migration of Siberian Barn Swallows Hirundo rustica tytleri. Journal of Ornithology, 167, 293–297. https://doi.org/10.1007/s10336-025-02306-z
Beck, H. E., McVicar, T. R., Vergopolan, N., Berg, A., Lutsko, N. J., Dufour, A., Zeng, Z., Jiang, X., van Dijk, A. I. J. M. & Miralles, D. G. (2023). High-resolution (1 km) Köppen-Geiger maps for 1901–2099 based on constrained CMIP6 projections. Scientific Data, 10(1), 724. https://doi.org/10.1038/s41597-023-02549-6
Bridle, J. & Hoffmann, A. (2022). Understanding the biology of species’ ranges: when and how does evolution change the rules of ecological engagement? Philosophical Transactions of the Royal Society B, 377(1848), 20210027. https://doi.org/10.1098/rstb.2021.0027
Brown, M. B. & Brown, C. R. (2020). Barn Swallow (Hirundo rustica), version 1.0. In: Rodewald, P. G. (ed.), Birds of the World. Cornell Lab of Ornithology, Ithaca, NY, USA. https://doi.org/10.2173/bow.barswa.01
Devi, M. N. & Devi, W. S. (2025). Meitei Society: A Historical and Socio-Cultural Exploration. International Journal of Humanities, Social Science and Management, 5(3), 814–821.
Dor, R., Safran, R. J., Sheldon, F. H., Winkler, D. W. & Lovette, I. J. (2010). Phylogeny of the genus Hirundo and the Barn Swallow subspecies complex. Molecular Phylogenetics and Evolution, 56(1), 409–418. https://doi.org/10.1016/j.ympev.2010.02.008
Fawthrop, R., Cerca, J., Pacheco, G., Sætre, G.-P., Scordato, E. S. C., Ravinet, M. & Rowe, M. (2025). Understanding human-commensalism through an ecological and evolutionary framework. Trends in Ecology & Evolution, 40(2), 159–169. https://doi.org/10.1016/j.tree.2024.10.006
Gaston, K. J. & Fuller, R. A. (2009). The sizes of species’ geographic ranges. Journal of Applied Ecology, 46(1), 1–9. https://doi.org/10.1111/j.1365-2664.2008.01596.x
Heim, W., Heim, R. J., Beermann, I., Burkovskiy, O. A., Gerasimov, Y., Ktitorov, P., Ozaki, K., Panov, I., Sander, M. M. et al. (2020). Using geolocator tracking data and ringing archives to validate citizen-science based seasonal predictions of bird distribution in a data-poor region. Global Ecology and Conservation, 24, e01215. https://doi.org/10.1016/j.gecco.2020.e01215
Hobson, K. A., Kardynal, K. J., Van Wilgenburg, S. L., Albrecht, G., Salvadori, A., Cadman, M. D., Liechti, F. & Fox, J. W. (2015). A continent-wide migratory divide in North American breeding Barn Swallows (Hirundo rustica). PloS One, 10(6), e0129340. https://doi.org/10.1371/journal.pone.0129340
Liechti, F., Scandolara, C., Rubolini, D., Ambrosini, R., Korner‐Nievergelt, F., Hahn, S., Lardelli, R., Romano, M., Caprioli, M. et al. (2015). Timing of migration and residence areas during the non‐breeding period of barn swallows Hirundo rustica in relation to sex and population. Journal of Avian Biology, 46(3), 254–265. https://doi.org/10.1111/jav.00485
Lombardo, G., Rambaldi Migliore, N., Colombo, G., Capodiferro, M. R., Formenti, G., Caprioli, M., Moroni, E., Caporali, L., Lancioni, H. & Secomandi, S. (2022). The mitogenome relationships and phylogeography of barn swallows (Hirundo rustica). Molecular Biology and Evolution, 39(6), msac113. https://doi.org/10.1093/molbev/msac113
Massimino, D., Johnston, A. & Pearce-Higgins, J. W. (2015). The geographical range of British birds expands during 15 years of warming. Bird Study, 62(4), 523–534. https://doi.org/10.1080/00063657.2015.1089835
Parmesan, C., Gaines, S., Gonzalez, L., Kaufman, D. M., Kingsolver, J., Townsend Peterson, A. & Sagarin, R. (2005). Empirical perspectives on species borders: from traditional biogeography to global change. Oikos, 108(1), 58–75. https://doi.org/10.1111/j.0030-1299.2005.13150.x
Pigot, A. L., Owens, I. P. F. & Orme, C. D. L. (2010). The environmental limits to geographic range expansion in birds. Ecology Letters, 13(6), 705–715. https://doi.org/10.1111/j.1461-0248.2010.01462.x
Rodríguez, J. P. (2002). Range contraction in declining North American bird populations. Ecological Applications, 12(1), 238–248. https://doi.org/10.1890/1051-0761(2002)012[0238:RCIDNA]2.0.CO;2
Scordato, E. S. C. & Safran, R. J. (2014). Geographic variation in sexual selection and implications for speciation in the Barn Swallow. Avian Research, 5(1). https://doi.org/10.1186/s40657-014-0008-4
Scordato, E. S. C., Smith, C. C. R., Semenov, G. A., Liu, Y., Wilkins, M. R., Liang, W., Rubtsov, A., Sundev, G., Koyama, K. et al. (2020). Migratory divides coincide with reproductive barriers across replicated avian hybrid zones above the Tibetan Plateau. Ecology Letters, 23(2), 231–241. https://doi.org/10.1111/ele.13420
Scordato, E. S. C., Wilkins, M. R., Semenov, G., Rubtsov, A. S., Kane, N. C. & Safran, R. J. (2017). Genomic variation across two barn swallow hybrid zones reveals traits associated with divergence in sympatry and allopatry. Molecular Ecology, 26(20), 5676–5691. https://doi.org/10.1111/mec.14276
Smith, C. C. R., Flaxman, S. M., Scordato, E. S. C., Kane, N. C., Hund, A. K., Sheta, B. M. & Safran, R. J. (2018). Demographic inference in barn swallows using whole‐genome data shows signal for bottleneck and subspecies differentiation during the Holocene. Molecular Ecology, 27(21), 4200–4212. https://doi.org/10.1111/mec.14854
Turbek, S. P., Schield, D. R., Scordato, E. S. C., Contina, A., Da, X.-W., Liu, Y., Liu, Y., Pagani-Núñez, E., Ren, Q.-M. & Smith, C. C. R. (2022). A migratory divide spanning two continents is associated with genomic and ecological divergence. Evolution, 76(4), 722–736. https://doi.org/10.1111/evo.14448
Turner, A. K. (2006). The barn swallow. A&C Black.
Turner, A. & Rose, C. (2010). A handbook to the swallows and martins of the world. A&C Black.
Winkler, D. W., Gandoy, F. A., Areta, J. I., Iliff, M. J., Rakhimberdiev, E., Kardynal, K. J., & Hobson, K. A. (2017). Long-distance range expansion and rapid adjustment of migration in a newly established population of barn swallows breeding in Argentina. Current Biology, 27(7), 1080–1084. https://doi.org/10.1016/j.cub.2017.03.006
Zink, R. M., Pavlova, A., Rohwer, S. & Drovetski, S. V. (2006). Barn swallows before barns: population histories and intercontinental colonization. Proceedings of the Royal Society B: Biological Sciences, 273(1591), 1245–1251. https://doi.org/10.1098/rspb.2005.3414
Edited By
Gopi G.V.
Wildlife Institute of India, Dehradun, India.
*CORRESPONDENCE
R Suresh Kumar
✉ suresh.wii@gmail.com
CITATION
Kaur, A. & Kumar, R. S. (2026). Barn Swallows of the Imphal Valley – a potential case of past climatic events leading to year-round residency in the population in Northeast India. Journal of Wildlife Science, 3(1), 35-40. https://doi.org/10.63033/JWLS.GXZU3002
FUNDING
This research was supported by the PhD Fellowship awarded to Amarjeet Kaur under the DST-INSPIRE scheme of the Government of India (No. DST/INSPIRE Fellowship/2018/IF180199).
COPYRIGHT
© 2026 Kaur & Kumar. This is an open-access article, immediately and freely available to read, download, and share. The information contained in this article is distributed under the terms of the Creative Commons Attribution License (CC BY 4.0), allowing for unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited in accordance with accepted academic practice. Copyright is retained by the author(s).
PUBLISHED BY
Wildlife Institute of India, Dehradun, 248 001 INDIA
PUBLISHER'S NOTE
The Publisher, Journal of Wildlife Science or Editors cannot be held responsible for any errors or consequences arising from the use of the information contained in this article. All claims expressed in this article are solely those of the author(s) and do not necessarily represent those of their affiliated organisations or those of the publisher, the editors and the reviewers. Any product that may be evaluated or used in this article or claim made by its manufacturer is not guaranteed or endorsed by the publisher.
Ali, S. & Ripley, S. D. (1987). Handbook of the Birds of India and Pakistan. Volume 5 Larks to the Grey Hypocolius. Oxford University Press.
Anisimova, V., Anisimov, Y., Bastardot, M., Beermann, I., Kunz, F. & Heim, W. (2026). Geolocator tracking and ring recoveries reveal the migration of Siberian Barn Swallows Hirundo rustica tytleri. Journal of Ornithology, 167, 293–297. https://doi.org/10.1007/s10336-025-02306-z
Beck, H. E., McVicar, T. R., Vergopolan, N., Berg, A., Lutsko, N. J., Dufour, A., Zeng, Z., Jiang, X., van Dijk, A. I. J. M. & Miralles, D. G. (2023). High-resolution (1 km) Köppen-Geiger maps for 1901–2099 based on constrained CMIP6 projections. Scientific Data, 10(1), 724. https://doi.org/10.1038/s41597-023-02549-6
Bridle, J. & Hoffmann, A. (2022). Understanding the biology of species’ ranges: when and how does evolution change the rules of ecological engagement? Philosophical Transactions of the Royal Society B, 377(1848), 20210027. https://doi.org/10.1098/rstb.2021.0027
Brown, M. B. & Brown, C. R. (2020). Barn Swallow (Hirundo rustica), version 1.0. In: Rodewald, P. G. (ed.), Birds of the World. Cornell Lab of Ornithology, Ithaca, NY, USA. https://doi.org/10.2173/bow.barswa.01
Devi, M. N. & Devi, W. S. (2025). Meitei Society: A Historical and Socio-Cultural Exploration. International Journal of Humanities, Social Science and Management, 5(3), 814–821.
Dor, R., Safran, R. J., Sheldon, F. H., Winkler, D. W. & Lovette, I. J. (2010). Phylogeny of the genus Hirundo and the Barn Swallow subspecies complex. Molecular Phylogenetics and Evolution, 56(1), 409–418. https://doi.org/10.1016/j.ympev.2010.02.008
Fawthrop, R., Cerca, J., Pacheco, G., Sætre, G.-P., Scordato, E. S. C., Ravinet, M. & Rowe, M. (2025). Understanding human-commensalism through an ecological and evolutionary framework. Trends in Ecology & Evolution, 40(2), 159–169. https://doi.org/10.1016/j.tree.2024.10.006
Gaston, K. J. & Fuller, R. A. (2009). The sizes of species’ geographic ranges. Journal of Applied Ecology, 46(1), 1–9. https://doi.org/10.1111/j.1365-2664.2008.01596.x
Heim, W., Heim, R. J., Beermann, I., Burkovskiy, O. A., Gerasimov, Y., Ktitorov, P., Ozaki, K., Panov, I., Sander, M. M. et al. (2020). Using geolocator tracking data and ringing archives to validate citizen-science based seasonal predictions of bird distribution in a data-poor region. Global Ecology and Conservation, 24, e01215. https://doi.org/10.1016/j.gecco.2020.e01215
Hobson, K. A., Kardynal, K. J., Van Wilgenburg, S. L., Albrecht, G., Salvadori, A., Cadman, M. D., Liechti, F. & Fox, J. W. (2015). A continent-wide migratory divide in North American breeding Barn Swallows (Hirundo rustica). PloS One, 10(6), e0129340. https://doi.org/10.1371/journal.pone.0129340
Liechti, F., Scandolara, C., Rubolini, D., Ambrosini, R., Korner‐Nievergelt, F., Hahn, S., Lardelli, R., Romano, M., Caprioli, M. et al. (2015). Timing of migration and residence areas during the non‐breeding period of barn swallows Hirundo rustica in relation to sex and population. Journal of Avian Biology, 46(3), 254–265. https://doi.org/10.1111/jav.00485
Lombardo, G., Rambaldi Migliore, N., Colombo, G., Capodiferro, M. R., Formenti, G., Caprioli, M., Moroni, E., Caporali, L., Lancioni, H. & Secomandi, S. (2022). The mitogenome relationships and phylogeography of barn swallows (Hirundo rustica). Molecular Biology and Evolution, 39(6), msac113. https://doi.org/10.1093/molbev/msac113
Massimino, D., Johnston, A. & Pearce-Higgins, J. W. (2015). The geographical range of British birds expands during 15 years of warming. Bird Study, 62(4), 523–534. https://doi.org/10.1080/00063657.2015.1089835
Parmesan, C., Gaines, S., Gonzalez, L., Kaufman, D. M., Kingsolver, J., Townsend Peterson, A. & Sagarin, R. (2005). Empirical perspectives on species borders: from traditional biogeography to global change. Oikos, 108(1), 58–75. https://doi.org/10.1111/j.0030-1299.2005.13150.x
Pigot, A. L., Owens, I. P. F. & Orme, C. D. L. (2010). The environmental limits to geographic range expansion in birds. Ecology Letters, 13(6), 705–715. https://doi.org/10.1111/j.1461-0248.2010.01462.x
Rodríguez, J. P. (2002). Range contraction in declining North American bird populations. Ecological Applications, 12(1), 238–248. https://doi.org/10.1890/1051-0761(2002)012[0238:RCIDNA]2.0.CO;2
Scordato, E. S. C. & Safran, R. J. (2014). Geographic variation in sexual selection and implications for speciation in the Barn Swallow. Avian Research, 5(1). https://doi.org/10.1186/s40657-014-0008-4
Scordato, E. S. C., Smith, C. C. R., Semenov, G. A., Liu, Y., Wilkins, M. R., Liang, W., Rubtsov, A., Sundev, G., Koyama, K. et al. (2020). Migratory divides coincide with reproductive barriers across replicated avian hybrid zones above the Tibetan Plateau. Ecology Letters, 23(2), 231–241. https://doi.org/10.1111/ele.13420
Scordato, E. S. C., Wilkins, M. R., Semenov, G., Rubtsov, A. S., Kane, N. C. & Safran, R. J. (2017). Genomic variation across two barn swallow hybrid zones reveals traits associated with divergence in sympatry and allopatry. Molecular Ecology, 26(20), 5676–5691. https://doi.org/10.1111/mec.14276
Smith, C. C. R., Flaxman, S. M., Scordato, E. S. C., Kane, N. C., Hund, A. K., Sheta, B. M. & Safran, R. J. (2018). Demographic inference in barn swallows using whole‐genome data shows signal for bottleneck and subspecies differentiation during the Holocene. Molecular Ecology, 27(21), 4200–4212. https://doi.org/10.1111/mec.14854
Turbek, S. P., Schield, D. R., Scordato, E. S. C., Contina, A., Da, X.-W., Liu, Y., Liu, Y., Pagani-Núñez, E., Ren, Q.-M. & Smith, C. C. R. (2022). A migratory divide spanning two continents is associated with genomic and ecological divergence. Evolution, 76(4), 722–736. https://doi.org/10.1111/evo.14448
Turner, A. K. (2006). The barn swallow. A&C Black.
Turner, A. & Rose, C. (2010). A handbook to the swallows and martins of the world. A&C Black.
Winkler, D. W., Gandoy, F. A., Areta, J. I., Iliff, M. J., Rakhimberdiev, E., Kardynal, K. J., & Hobson, K. A. (2017). Long-distance range expansion and rapid adjustment of migration in a newly established population of barn swallows breeding in Argentina. Current Biology, 27(7), 1080–1084. https://doi.org/10.1016/j.cub.2017.03.006
Zink, R. M., Pavlova, A., Rohwer, S. & Drovetski, S. V. (2006). Barn swallows before barns: population histories and intercontinental colonization. Proceedings of the Royal Society B: Biological Sciences, 273(1591), 1245–1251. https://doi.org/10.1098/rspb.2005.3414




